 Canwell Tibial Nail Intramedullary Nail Interlocking Cannualted Nail Canetn Gamma Nail
Canwell Tibial Nail Intramedullary Nail Interlocking Cannualted Nail Canetn Gamma Nail
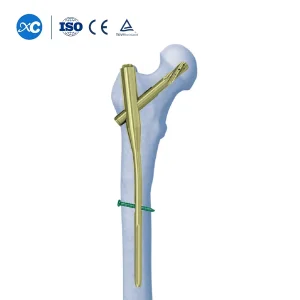 Price Gama Nails Pfna Surgery Nail Antirotation Titanium Femur Intramedullary Nail
Price Gama Nails Pfna Surgery Nail Antirotation Titanium Femur Intramedullary Nail
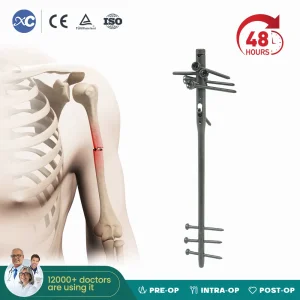 Xc Medico Orthopedic Implant Titanium Interlocking Humerus Nail Multi-Lock Humeral Intramedullary Nail
Xc Medico Orthopedic Implant Titanium Interlocking Humerus Nail Multi-Lock Humeral Intramedullary Nail
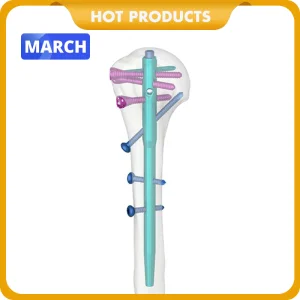 Medical Supply Orthopedic Implant Titanium Interlocking Humerus Nail Multi-Lock Humeral Intramedullary Nail
Medical Supply Orthopedic Implant Titanium Interlocking Humerus Nail Multi-Lock Humeral Intramedullary Nail
 Orthopedic Implant Elastic Intramedullary Nail, Orthopedic Implant Ulna Radius Intramedullary Nail
Orthopedic Implant Elastic Intramedullary Nail, Orthopedic Implant Ulna Radius Intramedullary Nail
 Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail
Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail
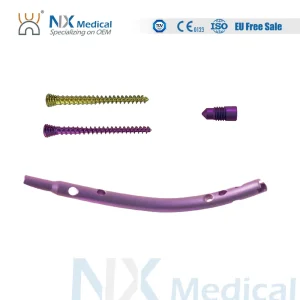 Nx Medical Retrograde Tibial Nail Orthopedic Implant Intramedullary Nail for Internal Fixation
Nx Medical Retrograde Tibial Nail Orthopedic Implant Intramedullary Nail for Internal Fixation
 High-Quality Interlocking Nail Intramedullary Nail in Blue Green Gold Colors
High-Quality Interlocking Nail Intramedullary Nail in Blue Green Gold Colors
The Gamma Nail System has revolutionized the treatment of trochanteric fractures, particularly in the aging global population. As medical science advances, the demand for intramedullary nails—specifically the Gamma and PFNA (Proximal Femoral Nail Antirotation) types—has surged. Globally, the orthopedic trauma fixation market is expanding at a CAGR of over 5%, driven by an increasing incidence of osteoporotic fractures in developed nations and rising trauma cases in developing economies.
In the current industrial landscape, the Gamma Nail System is considered the "gold standard" for stable and unstable peritrochanteric fractures. Surgeons worldwide prefer these systems due to their biomechanical advantages: a shorter lever arm compared to sliding hip screws, better load-sharing properties, and a minimally invasive approach that significantly reduces surgical blood loss and post-operative recovery time.
Development of hydroxyapatite and antibacterial coatings to prevent implant-related infections and promote osseointegration.
Exploring radiolucent materials that offer elasticity closer to natural bone while allowing clearer post-op imaging.
Integration with surgical robotics and AI-guided distal locking systems for pinpoint accuracy in complex trauma cases.
The industry is shifting towards precision medicine. Modern Gamma Nail systems now incorporate anatomic designs that account for the natural curvature of the femur, reducing the risk of anterior cortex perforation. As a leading manufacturer in China, we are at the forefront of these trends, utilizing CNC precision machining and advanced surface treatment technologies to ensure our implants meet the rigorous standards of global healthcare providers.
Foshan Wigivida Medical Co., Ltd. is a leading China-based manufacturer of high-quality medical consumables and devices, specializing in respiratory products, medical tubes, urology products, hypodermic and surgical consumables. With years of dedicated experience in the healthcare industry, Wigivida has built a reputation for providing reliable, safe, and innovative solutions to hospitals, clinics, and medical distributors worldwide.
The company offers a comprehensive product range, including oxygen masks, ventilator accessories, catheters, Foley catheters, hypodermic needles, syringes, surgical drapes, sterile gloves, sutures, cotton swabs, gauze rolls, IV sets, and disposable medical kits. Each product is manufactured with strict adherence to quality standards, ensuring biocompatibility, durability, and patient safety.


Wigivida Medical emphasizes innovation, research, and precision manufacturing, combining advanced production technology with a rigorous quality management system. The company also provides customized solutions to meet the specific needs of healthcare providers, supporting efficient hospital workflows and minimizing infection risks.
Guided by the principles of "quality first, service first, and customer satisfaction-driven," Foshan Wigivida Medical Co., Ltd. has earned an excellent reputation both domestically and internationally. With a professional R&D team, modern manufacturing facilities, and dedicated after-sales service, the company continues to advance the medical consumables industry, delivering safe, effective, and high-quality healthcare solutions.
Procurement departments in modern hospitals and international distributors focus on three pillars: Quality, Certifications (CE/FDA/ISO), and Cost-Efficiency. China's Gamma Nail system manufacturers have bridged the gap between high-end performance and affordable pricing. By leveraging a robust supply chain, we offer titanium alloy (Ti6Al4V ELI) implants that are biocompatible and MRI-safe at a fraction of the cost of Western counterparts.
As a specialized exporter, we provide not just the implant, but the complete instrumentation set. Our surgical kits are designed for ergonomic use, featuring color-coded drills and guides that simplify the surgical workflow. We offer comprehensive OEM/ODM services, allowing distributors to customize sizing and packaging to meet local regulatory requirements in Europe, Southeast Asia, and Latin America.
 Orthopedic Implant Interlocking Nail Standard Intertan Intramedullary Nail for Fracture Surgery
Orthopedic Implant Interlocking Nail Standard Intertan Intramedullary Nail for Fracture Surgery
 Orthopedic Implant Interlocking Nail Standard Intertan Intramedullary Nail for Fracture Surgery
Orthopedic Implant Interlocking Nail Standard Intertan Intramedullary Nail for Fracture Surgery
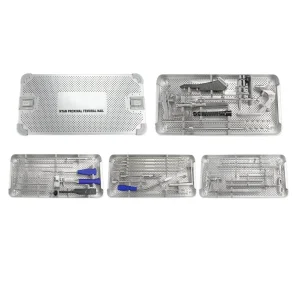 Orthopedic Ntan Proximal Femoral Nail Medical Equipment Surgical Instrument
Orthopedic Ntan Proximal Femoral Nail Medical Equipment Surgical Instrument
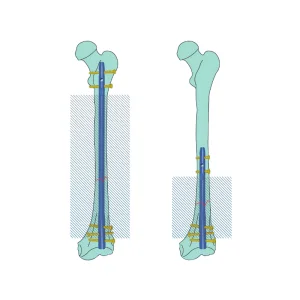 Czmeditech Orthopedic Expert Reversed Femoral Intramedullary Nail
Czmeditech Orthopedic Expert Reversed Femoral Intramedullary Nail
 Surgical Instrument Femoral Interlocking Nail Instrument Set for Orthopedic Bone Surgery
Surgical Instrument Femoral Interlocking Nail Instrument Set for Orthopedic Bone Surgery
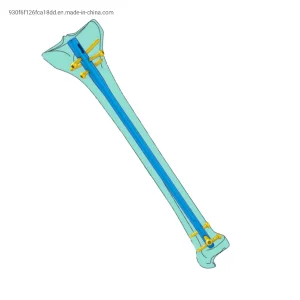 Intramedullary Nail System Expert Tibial Interlocking Nail Suprapatellar Approach
Intramedullary Nail System Expert Tibial Interlocking Nail Suprapatellar Approach
 Orthopedic Surgical Instrument Medical Femoral Interlocking Nail Instrument Set
Orthopedic Surgical Instrument Medical Femoral Interlocking Nail Instrument Set
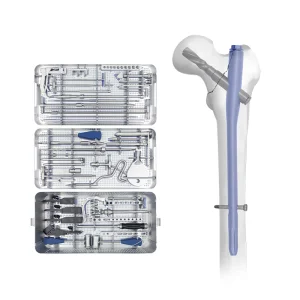 Medical Supply Orthopedic Implant Titanium Femur Pfna Intramedullary Nail Instrument Set
Medical Supply Orthopedic Implant Titanium Femur Pfna Intramedullary Nail Instrument Set
Selecting a Gamma Nail system involves a deep understanding of biomechanical stability. As experienced manufacturers, we emphasize the importance of the nail-lag screw interface. Our latest designs include an "anti-rotation" feature that prevents the "Z-effect," a common complication in femoral fracture fixation. By utilizing high-strength titanium alloys, our nails offer the perfect balance of stiffness and flexibility, mimicking the natural modulus of human bone to prevent stress shielding.
Our commitment to E-E-A-T (Experience, Expertise, Authoritativeness, and Trustworthiness) means we don't just sell products; we provide clinical support. Our R&D department works closely with orthopedic surgeons to refine our instrument sets, ensuring that distal locking is achieved quickly even without advanced imaging, thus reducing radiation exposure for the surgical team. Whether you are a hospital procurement officer in Brazil or a distributor in Germany, our China-based manufacturing hub provides the reliability and technical excellence required for modern trauma surgery.