 Headless Compression Hollow Screw Cannulated Screw Bone Screws Orthopedic Implants
Headless Compression Hollow Screw Cannulated Screw Bone Screws Orthopedic Implants
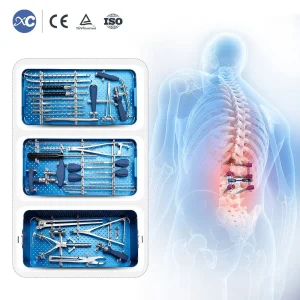 Orthopedic Bone Surgical Set 6.0mm Spinal Pedicle Screw System Instrument Set
Orthopedic Bone Surgical Set 6.0mm Spinal Pedicle Screw System Instrument Set
 Trauma Implant Micro Straight Maxillofacial Trauma Plate 1.5mm Screw
Trauma Implant Micro Straight Maxillofacial Trauma Plate 1.5mm Screw
 Orthopedic Titanium Pedicle Screw Instrument Set 6.0mm Spinal Pedicle Screw
Orthopedic Titanium Pedicle Screw Instrument Set 6.0mm Spinal Pedicle Screw
 Surgery Orthopedic Implants Cannulated Locking Compression Screw
Surgery Orthopedic Implants Cannulated Locking Compression Screw
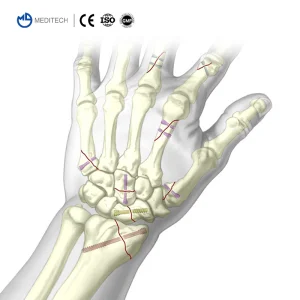 Titanium Medical Bone Screws Herbert Cannulated Compression Screw Price
Titanium Medical Bone Screws Herbert Cannulated Compression Screw Price
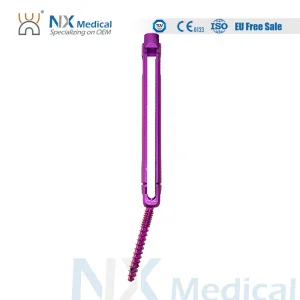 Nx Medical Titanium Cannulated Bone Cement Pedicle Screw OEM ODM
Nx Medical Titanium Cannulated Bone Cement Pedicle Screw OEM ODM
 Orthopedic Maxillofacial Micro Straight Plate Titanium Implant
Orthopedic Maxillofacial Micro Straight Plate Titanium Implant
The global orthopedic implant market is witnessing a significant surge, with Cancellous Bone Screws playing a pivotal role in trauma and reconstructive surgeries. Unlike cortical screws, cancellous screws are designed with deeper threads and a larger pitch to secure fixation in spongy, porous bone. Today, the demand is driven by an aging global population and a rise in sports-related injuries.
As a leading China Cancellous Bone Screw factory, we analyze the shifting trends: North America and Europe remain the largest markets due to advanced healthcare infrastructure, but the Asia-Pacific region is the fastest-growing. Hospitals and surgical centers worldwide are increasingly seeking reliable exporters who can provide CE and ISO certified implants that balance surgical precision with cost-effectiveness.

Transition from traditional stainless steel to Grade 5 Titanium and PEEK (Polyether ether ketone) for better MRI compatibility and reduced stress shielding.
Utilizing AI-driven Swiss-type CNC lathes to ensure thread accuracy within microns, crucial for headless compression and cannulated screw designs.
Advanced anodic oxidation and sandblasting techniques to enhance osseointegration, ensuring the screw integrates seamlessly with the patient's bone structure.
Implementation of UDI (Unique Device Identification) for global traceability, meeting the stringent requirements of the FDA and EU MDR.
International medical distributors and hospital procurement departments face the challenge of sourcing high-quality implants that meet local regulatory standards while managing costs. Our role as an exporter of cancellous bone screws involves more than just shipping products; it encompasses comprehensive quality assurance.
We provide localized documentation support, customized packaging, and sterile vs. non-sterile options. Whether it's for metaphyseal fractures in Brazil or joint replacements in Germany, our products are engineered to withstand the mechanical stresses required for various localized application scenarios.

Foshan Wigivida Medical Co., Ltd. is a leading China-based manufacturer of high-quality medical consumables and devices, specializing in respiratory products, medical tubes, urology products, hypodermic and surgical consumables. With years of dedicated experience in the healthcare industry, Wigivida has built a reputation for providing reliable, safe, and innovative solutions to hospitals, clinics, and medical distributors worldwide.
The company offers a comprehensive product range, including oxygen masks, ventilator accessories, catheters, Foley catheters, hypodermic needles, syringes, surgical drapes, sterile gloves, sutures, cotton swabs, gauze rolls, IV sets, and disposable medical kits. Each product is manufactured with strict adherence to quality standards, ensuring biocompatibility, durability, and patient safety.
Wigivida Medical emphasizes innovation, research, and precision manufacturing, combining advanced production technology with a rigorous quality management system. Guided by the principles of "quality first, service first, and customer satisfaction-driven," we have earned an excellent reputation both domestically and internationally. Our professional R&D team and modern manufacturing facilities allow us to stay at the forefront of the orthopedic and medical consumable industry.
Cancellous bone screws are utilized in a variety of critical surgical procedures. As a factory with deep expertise, we understand the nuances of these applications:
Used for the fixation of fractures in epiphysis and metaphysis of long bones where cancellous bone is predominant. Our screws provide superior compression to facilitate faster healing.
Essential for ACL/PCL reconstructions. Our PEEK and Titanium interference screws offer the high pull-out strength required for ligament reattachment.
In osteoporotic patients, cannulated cancellous screws with bone cement augmentation options are used to ensure stable pedicle fixation.
 Non-Magnetic 316L Stainless Steel Standoff Screw for MRI Room Equipment
Non-Magnetic 316L Stainless Steel Standoff Screw for MRI Room Equipment
 Acl Interference Screw for Knee Arthroscopy Peek Screw Set
Acl Interference Screw for Knee Arthroscopy Peek Screw Set
 Stainless Steel Bone Fixator for Orthopedic Surgery
Stainless Steel Bone Fixator for Orthopedic Surgery
 Insulation Peek Screw Hex Head Plastic Screw DIN 912
Insulation Peek Screw Hex Head Plastic Screw DIN 912
 Medical Spine Bone Cement Cannulated Pedicle Screw
Medical Spine Bone Cement Cannulated Pedicle Screw
 Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System
Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System
 Variable Angle Distal Volar Radius Compression LCP Bone Plate
Variable Angle Distal Volar Radius Compression LCP Bone Plate
 Nx Medical Titanium Cannulated Bone Cement Pedicle Screw Internal
Nx Medical Titanium Cannulated Bone Cement Pedicle Screw Internal
Cancellous bone screws are specifically engineered for the anatomy of "spongy" bone. The design features a larger outer diameter and a deeper thread profile compared to cortical screws. This allows the screw to grip the trabecular network of the bone effectively. As a specialized Cancellous Bone Screw factory, we offer both fully threaded and partially threaded versions. Partially threaded screws are particularly effective for lag fixation, where compression is required across a fracture line.
In the global market, Titanium Alloy (Ti-6Al-4V ELI) has become the gold standard due to its excellent biocompatibility and high strength-to-weight ratio. It also reduces interference during postoperative imaging (MRI and CT scans). However, 316L Stainless Steel remains a preferred choice for certain temporary fixations due to its cost-efficiency and high fatigue strength. Our factory maintains stocks of both materials to meet diverse global clinical requirements.
The introduction of cannulated cancellous bone screws has revolutionized minimally invasive surgery (MIS). By allowing the screw to be inserted over a guide wire, surgeons can achieve much higher precision in placement, especially in complex areas like the femoral neck or the scaphoid. Our manufacturing process ensures the central lumen is perfectly concentric, preventing guide wire binding and ensuring smooth insertion.
China's medical device manufacturing sector has undergone a quality revolution. By combining high-volume production capabilities with stringent international standards, factories like ours provide a competitive edge in the global supply chain. We utilize multi-axis machining centers that allow for the production of complex geometries in a single setup, reducing human error and ensuring that every batch of screws meets the ASTM F136 or ISO 5832-3 standards for surgical implants.
The industry is moving toward bio-absorbable materials that disappear once the bone has healed, eliminating the need for a second surgery to remove the implant. Additionally, research into "smart" coatings that release antimicrobial agents to prevent surgical site infections is gaining momentum. We are actively investing in R&D to bring these future-proof technologies to our international clients, cementing our position as a top-tier exporter.
Choosing a Cancellous Bone Screw exporter involves evaluating their ability to navigate complex international trade regulations. We offer end-to-end logistics solutions, from sterile packaging in Class 100,000 cleanrooms to customs clearance assistance. Our goal is to ensure that healthcare providers have the tools they need to save lives and improve patient mobility, regardless of their location.