
Office Chair Rollerblade Swivel Wheel Casters Total Lock Replacement Chair Casters

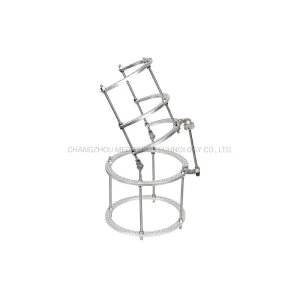
Ilizarov Ring External Fixator Orthopedic Instruments Wrist Joint External Fixator


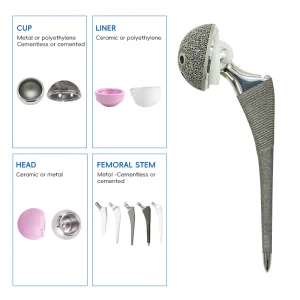
Sports Medicine Tibial Ligament Reconstruction Implants Peek Interference Screw


Orthofix Design Three-Section Straight Type External Fixator for Orthopedic Fixation
The global medical landscape is witnessing a seismic shift towards Minimally Invasive Surgery (MIS). At the heart of this revolution lies the Hollow Drill Bit, also known as the cannulated drill. These precision-engineered tools are indispensable for orthopedic trauma surgeries, allowing surgeons to insert guidewires and subsequently place cannulated screws with sub-millimeter accuracy. As China cements its position as a global manufacturing powerhouse, top factories are no longer just "suppliers"—they are strategic R&D partners for international healthcare distributors.
Hospitals in North America and Europe are increasingly seeking "Information Gain" in their supply chains, prioritizing factories that offer transparent material sourcing (such as Grade 5 Titanium) and ISO 13485 compliance. The demand is driven by the rise in geriatric fractures and sports medicine interventions.
Modern factories are integrating vertical manufacturing—from raw titanium slitting to final laser marking. This ensures a "Single Source of Truth" for quality control, reducing the risk of surgical site infections (SSIs) through superior surface finishing and edge retention.
The industrialization of hollow drill bit production in China has reduced the global cost of orthopedic trauma kits by approximately 30-40%, making advanced surgical procedures accessible in emerging markets across Southeast Asia and Latin America.
Foshan Wigivida Medical Co., Ltd. is a leading China-based manufacturer of high-quality medical consumables and devices, specializing in respiratory products, medical tubes, urology products, hypodermic and surgical consumables. With years of dedicated experience in the healthcare industry, Wigivida has built a reputation for providing reliable, safe, and innovative solutions to hospitals, clinics, and medical distributors worldwide.
The company offers a comprehensive product range, including oxygen masks, ventilator accessories, catheters, Foley catheters, hypodermic needles, syringes, surgical drapes, sterile gloves, sutures, cotton swabs, gauze rolls, IV sets, and disposable medical kits. Each product is manufactured with strict adherence to quality standards, ensuring biocompatibility, durability, and patient safety.

Wigivida Medical emphasizes innovation, research, and precision manufacturing, combining advanced production technology with a rigorous quality management system. Guided by the principles of "quality first, service first, and customer satisfaction-driven," the company continues to deliver safe, effective, and high-quality healthcare solutions.

Transitioning from standard surgical steel to ultra-hard Stainless Steel 440 Mod and Titanium alloys to ensure the drill bit maintains sharpness through multiple sterilization cycles.
Utilizing 5-axis CNC Swiss-turning centers to achieve internal diameters (cannulation) with perfect concentricity, crucial for high-speed guidewire stability.
Implementing Titanium Nitride (TiN) or Diamond-Like Carbon (DLC) coatings to reduce friction-generated heat, which prevents bone necrosis during high-torque drilling.













Navigating the EU MDR (Medical Device Regulation) and US FDA 510(k) pathways is complex. Our engineering teams provide full technical dossiers and clinical evaluation reports (CER) to assist our exporters in local market entry.
By leveraging Foshan's logistical hub, we offer JIT (Just-In-Time) delivery and safety stock management for global distributors, ensuring that critical surgeries are never delayed due to instrument shortages.
We provide training modules for local sales representatives and hospital sterilization departments, ensuring our hollow drill bits are used and maintained according to international best practices.




Most premium hollow drill bits are manufactured from customized Stainless Steel 440 Mod or medical-grade Titanium alloys. Titanium offers superior biocompatibility, while specialized steel provides the high hardness necessary for multiple usages without dulling the cutting edge.
We utilize deep-hole drilling technology and Swiss-style CNC machines. Each batch undergoes 100% inspection using laser micrometers to ensure that the internal hollow channel is perfectly centered, preventing guidewire "wobble" during surgery.
Yes. As a leading exporter, we offer OEM/ODM services where the flute length, diameter, and connector type (e.g., AO quick-couple or Stryker-type) can be tailored to your existing instrument kits.
For the EU, CE marking under the MDR is mandatory. For the USA, FDA 510(k) clearance is typically required for orthopedic instruments. Our factory maintains ISO 13485 certification to support these regulatory requirements.



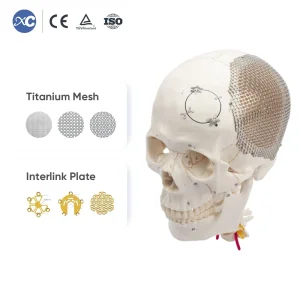
Orthopedic Maxillofacial Neurosurgery Surgical Titanium Cranial Mesh Implant

Orthopedic External Fixator - Ilizarov External Fixator-Half Ring-Alluminum Alloy
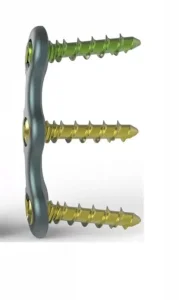
Anterior Cervical Fixation System with Screw for Cervical Spine with CE Certificate
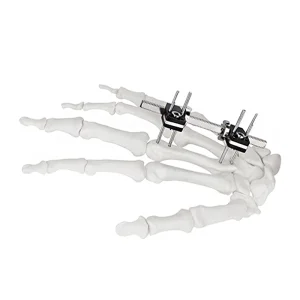
Cn Manufacturer Finger External Fixator for Mini Fixator Straight Type


