 5.5/6.5 Orthopedic Series Spinal System Pedicle Spinal Screws
5.5/6.5 Orthopedic Series Spinal System Pedicle Spinal Screws
 Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set
Orthopedic Surgical Instrument Spinal Fixation System 6.0 mm Pedicle Screw Instruments Set
 Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System Surgical Instrument Set
Xc Medico Orthopedic 6.0mm System Spinal Pedicle Screw System Surgical Instrument Set
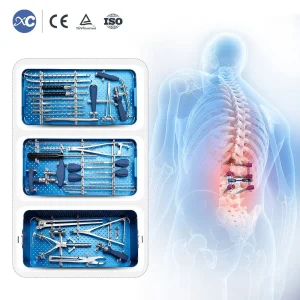 Orthopedic Bone Surgical Set 6.0mm Spinal Pedicle Screw System Instrument Set
Orthopedic Bone Surgical Set 6.0mm Spinal Pedicle Screw System Instrument Set
 5.5mm Spine Orthopedic Surgical Implants Surgery Pedicle Screw Instrument Set
5.5mm Spine Orthopedic Surgical Implants Surgery Pedicle Screw Instrument Set
 Orthopedic Spinal Fixation Medical Instrument Vertebral Hook
Orthopedic Spinal Fixation Medical Instrument Vertebral Hook
 Orthopedic Titanium Pedicle Screw Instrument Set 6.0mm Spinal Pedicle Screw Instrument Set
Orthopedic Titanium Pedicle Screw Instrument Set 6.0mm Spinal Pedicle Screw Instrument Set
 Orthopedic Spinal Fixation Medical Instrument Spine Vertebral Hook
Orthopedic Spinal Fixation Medical Instrument Spine Vertebral Hook
As a premier OEM/ODM Spinal Cross Link factory, Foshan Wigivida Medical Co., Ltd. stands at the intersection of surgical precision and industrial innovation. Spinal cross links, also known as transverse connectors, play a pivotal role in maintaining the structural integrity of pedicle screw constructs. By providing rotational stability and preventing rod migration, these components are essential for successful posterior spinal fusion.
Our expertise is rooted in a deep understanding of biomechanical engineering and clinical requirements. We leverage high-grade Titanium Alloy (Ti6Al4V) to manufacture cross links that offer the perfect balance of strength, biocompatibility, and low-profile design. For global distributors and hospital chains, sourcing from a specialized Chinese factory ensures access to cutting-edge technology backed by rigorous ISO 13485 and CE certifications.

The aging global population has led to an unprecedented increase in degenerative spinal disorders. Consequently, the demand for spinal fixation systems is projected to grow by 5.8% CAGR annually through 2030, with a focus on stable, long-term implant solutions.
Current industrial trends are shifting toward additive manufacturing (3D printing) and PEEK materials. Our R&D team continuously explores surface treatments that enhance osseointegration, ensuring our OEM partners stay ahead of the technology curve.
The rise of surgical robotics requires implants with higher dimensional accuracy. Our Spinal Cross Links are manufactured with precision tolerances (±0.01mm) to ensure seamless integration with the latest intraoperative navigation systems.
Global medical device procurement involves more than just cost-analysis; it requires a partner who understands the nuance of local medical regulations. Whether you are a distributor in Europe requiring MDR compliance or a surgical provider in Southeast Asia looking for cost-effective excellence, Wigivida provides a tailored approach.

Foshan Wigivida Medical Co., Ltd. is a leading China-based manufacturer of high-quality medical consumables and devices, specializing in respiratory products, medical tubes, urology products, hypodermic and surgical consumables. With years of dedicated experience in the healthcare industry, Wigivida has built a reputation for providing reliable, safe, and innovative solutions to hospitals, clinics, and medical distributors worldwide.
The company offers a comprehensive product range, including oxygen masks, ventilator accessories, catheters, Foley catheters, hypodermic needles, syringes, surgical drapes, sterile gloves, sutures, cotton swabs, gauze rolls, IV sets, and disposable medical kits. Each product is manufactured with strict adherence to quality standards, ensuring biocompatibility, durability, and patient safety.
Wigivida Medical emphasizes innovation, research, and precision manufacturing, combining advanced production technology with a rigorous quality management system. The company also provides customized solutions to meet the specific needs of healthcare providers, supporting efficient hospital workflows and minimizing infection risks.
Guided by the principles of "quality first, service first, and customer satisfaction-driven," Foshan Wigivida Medical Co., Ltd. has earned an excellent reputation both domestically and internationally. With a professional R&D team, modern manufacturing facilities, and dedicated after-sales service, the company continues to advance the medical consumables industry, delivering safe, effective, and high-quality healthcare solutions that improve patient outcomes and support global healthcare innovation.
The application of Spinal Cross Links varies significantly across clinical disciplines. In pediatric deformity correction (scoliosis), the cross link must be adjustable to accommodate growing spines. In trauma cases, high-strength rigid connectors are preferred to stabilize fractured vertebrae immediately.
Designed for rapid assembly in the OR, reducing anesthesia time for the patient and improving turnover for the hospital.
Our OEM packaging options allow local brands to offer "German-standard" quality with localized branding and documentation.
For complex revision surgeries, our team can provide custom-machined ODM components based on CT-scan data.
 Orthopedic Spinal Fixation Vertebral Hook Medical Instrument
Orthopedic Spinal Fixation Vertebral Hook Medical Instrument
 Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail
Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail
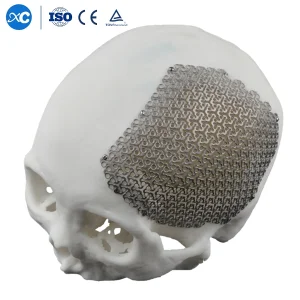 3D Anatomical Cloud Shape Cmf Cranial Mesh Titanium Maxillofacial Implant
3D Anatomical Cloud Shape Cmf Cranial Mesh Titanium Maxillofacial Implant
 Adjustable Shoulder Abduction Bracket for Enhanced Comfort and Support
Adjustable Shoulder Abduction Bracket for Enhanced Comfort and Support
 Adjustable Shoulder Abduction Bracket for Enhanced Comfort and Support
Adjustable Shoulder Abduction Bracket for Enhanced Comfort and Support
 Premium Shoulder Abduction Support Strap for Comfort and Mobility
Premium Shoulder Abduction Support Strap for Comfort and Mobility
 Versatile Shoulder Abduction Bracket for Pain Relief and Stability with CE
Versatile Shoulder Abduction Bracket for Pain Relief and Stability with CE
 Premium Adjustable Memory Foam Shoulder Pillow for Restful Sleep
Premium Adjustable Memory Foam Shoulder Pillow for Restful Sleep