

Medical Well-Leg Positioners Knee Platform Positioner / Total Knee Replacement / Tkr Positioner

China Factory Orthopedic Implants Prosthetic Knee/Knee Joint Prosthesis/Artificial Knee Replacement
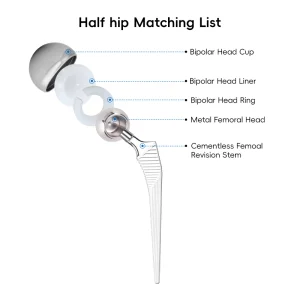
Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Total Knee Joint Replacement Instrument Set
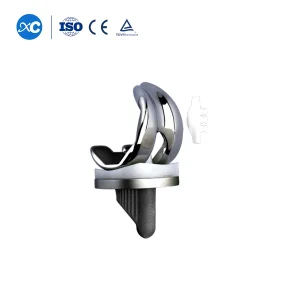
Tka/Tkr Primary Total Knee Joint Prosthesis Knee Orthopedic Implants Artificial Knee Replacement

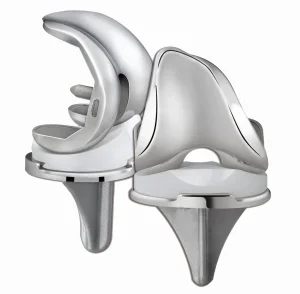
Orthopedic Implants Artificial Knee Joint Prosthesis for Total Replacement System
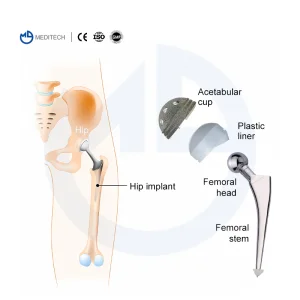
Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
The global orthopedic surgery landscape has shifted significantly towards the Posterior Stabilized (PS) Knee design. Unlike Cruciate Retaining (CR) models, PS knee systems are engineered to replace the function of the Posterior Cruciate Ligament (PCL) using a mechanical cam-and-post mechanism. This design is highly favored in complex primary Total Knee Arthroplasty (TKA) and revision surgeries where the patient's own ligaments are compromised.
Current industrial data suggests the orthopedic implant market is poised to reach USD 12 billion by 2028, with PS systems accounting for over 60% of total knee reconstructions in North America and Europe. This growth is driven by an aging global population, rising obesity rates, and the clinical success of high-flexion PS designs that offer superior range of motion and joint stability.
Modern PS knee systems are increasingly compatible with robotic-assisted surgery, allowing for sub-millimeter precision in bone resection and implant alignment, reducing revision rates by 30%.
The use of 3D-printed porous titanium for femoral and tibial components promotes biological osseointegration, providing a "cementless" solution that is ideal for younger, active patients.
Highly Cross-Linked Polyethylene (HXLPE) infused with Vitamin E is the new gold standard for PS knee inserts, dramatically reducing wear particles and inflammation.
The PS knee prosthesis utilizes a "Cam and Post" mechanism. During knee flexion, the femoral cam engages with the tibial post, forcing the femur to roll back on the tibia. This "femoral rollback" mimics the natural PCL function, preventing anterior subluxation and allowing for deeper flexion without mechanical impingement.
Key Advantage: PS designs simplify the surgical procedure because the surgeon does not need to balance a tight or degenerated PCL, making the outcome more predictable even in severe varus or valgus deformities.
At the core of a top-tier PS knee is Ti6Al4V Titanium Alloy or Cobalt-Chromium (CoCrMo). These materials offer the fatigue strength required to withstand millions of gait cycles. Our factory employs precision CNC machining to ensure the articular surfaces have a mirror-like finish, reducing the coefficient of friction to near-biological levels.

Foshan Wigivida Medical Co., Ltd. is a leading China-based manufacturer of high-quality medical consumables and devices, specializing in respiratory products, medical tubes, urology products, hypodermic and surgical consumables. With years of dedicated experience in the healthcare industry, Wigivida has built a reputation for providing reliable, safe, and innovative solutions to hospitals, clinics, and medical distributors worldwide.
Guided by the principles of "quality first, service first, and customer satisfaction-driven," Foshan Wigivida Medical Co., Ltd. has earned an excellent reputation both domestically and internationally. With a professional R&D team, modern manufacturing facilities, and dedicated after-sales service, the company continues to advance the medical consumables industry, delivering safe, effective, and high-quality healthcare solutions that improve patient outcomes and support global healthcare innovation.
The company offers a comprehensive product range, including oxygen masks, ventilator accessories, catheters, Foley catheters, hypodermic needles, syringes, surgical drapes, sterile gloves, sutures, cotton swabs, gauze rolls, IV sets, and disposable medical kits. Each product is manufactured with strict adherence to quality standards, ensuring biocompatibility, durability, and patient safety.

Wigivida Medical emphasizes innovation, research, and precision manufacturing, combining advanced production technology with a rigorous quality management system. The company also provides customized solutions to meet the specific needs of healthcare providers, supporting efficient hospital workflows and minimizing infection risks.
By leveraging China's mature industrial supply chain, we provide high-grade orthopedic implants at a fraction of the cost of Western competitors, without compromising on ASTM F136 titanium standards.
Our Foshan facility is equipped with automated CNC lines capable of producing thousands of units monthly, ensuring that large-scale tenders from government hospitals are met on time.
We adhere to ISO 13485 and CE MDD/MDR frameworks. Every PS knee undergoes rigorous fatigue testing (up to 5 million cycles) before leaving the factory.
Procurement managers from different regions have varying needs. In Western Europe and North America, the focus is on "Revision Compatibility" and "Cementless Options." In Southeast Asia and Latin America, the demand is for "Primary TKA Durability" and "Surgical Instrument Versatility."
As a global supplier, we provide localized surgical instrument sets that are intuitive for surgeons trained in different medical schools, ensuring a smooth transition to our PS knee system in any OR environment.

Orthopedic Surgical Medical Acl Surgery Arthroscopy Instrument Set for Knee Reconstruction

Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Artificial Replacement


Factory Price Total Knee Replacement Implant Knee Prosthesis Instrumental Arthroscopy Instrument

Orthopedic Toolmed Knee Prosthesis Manufacturer Knee Replacement Ortho Surgery Arthroplasty Tka

Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China
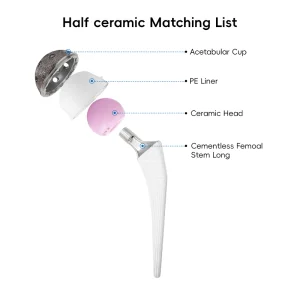
Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement
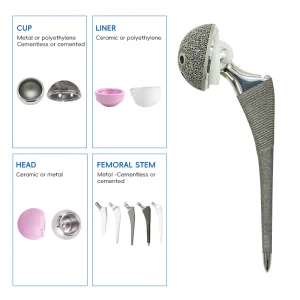
As we look toward 2030, the Top 10 Posterior Stabilized Knee Manufacturers are shifting from generic solutions to Data-Driven Orthopedics. By combining world-class manufacturing in Foshan with global clinical feedback, Wigivida Medical is not just a factory; we are a partner in restoring mobility to millions. Our commitment to high-performance polymers, titanium alloys, and precision instrumentation ensures that every TKA procedure leads to a better quality of life.