 Sports Medical Orthopedic Arthroscopy Implants Acl Reconstruction Peek Acl Bioabsorbable Interference Screw
Sports Medical Orthopedic Arthroscopy Implants Acl Reconstruction Peek Acl Bioabsorbable Interference Screw
 Grab Slotted Headless Plastic Flat Point End Bolt Plla Absorbable Interference Screw Screw
Grab Slotted Headless Plastic Flat Point End Bolt Plla Absorbable Interference Screw Screw
 Sports Medicine Tibial Ligament Reconstruction Implants Peek Interference Screw
Sports Medicine Tibial Ligament Reconstruction Implants Peek Interference Screw
 Surgical Instrument Acl Interference Screws, Canulated Screws II
Surgical Instrument Acl Interference Screws, Canulated Screws II
New Zealand has long been recognized for its world-class healthcare system and a population that is deeply engaged in high-impact sports, ranging from rugby and netball to mountain biking and skiing. This active lifestyle, while beneficial for public health, has led to a significant demand for advanced orthopedic interventions, particularly in the field of ACL (Anterior Cruciate Ligament) and PCL reconstruction. As a primary manufacturer and exporter, Yuna Medical provides high-precision interference screws that meet the rigorous standards of New Zealand’s District Health Boards (DHBs) and private surgical facilities like Southern Cross Hospitals.
In the current commercial climate of Auckland, Wellington, and Christchurch, there is a clear trend toward minimally invasive arthroscopic procedures. Surgeons are increasingly moving away from traditional metal fixation toward PEEK (Polyether ether ketone) and Bioabsorbable (PLLA/Biocomposite) materials. These technologies offer superior biocompatibility, reduced MRI interference, and a modular approach to patient recovery—factors that are highly prioritized by New Zealand’s medical practitioners.
• 15% increase in arthroscopic surgeries annually.
• High preference for PEEK materials in private clinics.
• ACC-funded rehabilitation driving high implant volume.
Surgeon Satisfaction Rate
Changzhou Yuna Medical Apparatus And Instruments Co., Ltd. was established in 1992. As a high-tech enterprise integrating research, development, manufacturing, and sales, we have maintained steady growth for over 30 years.

With 30+ years of experience, Yuna Medical continuously advances in orthopedic technology and global market presence, providing reliable supply chains for New Zealand importers.

Over 20% of employees are R&D professionals driving innovation in orthopedic and rehabilitation solutions, ensuring our interference screws remain at the cutting edge.

Certified by ISO, CE, FDA, and CCC, ensuring top-tier product quality and full international compliance for the New Zealand Medsafe regulatory framework.

Collaborating with leading universities and clinical experts globally to enhance research, product development, and surgical effectiveness in sports medicine.
The manufacturing of interference screws is currently undergoing a paradigm shift. In New Zealand, the focus has shifted toward Bio-composite materials that not only provide immediate mechanical stability but also facilitate bone ingrowth during the healing process. Our PLLA Absorbable Interference Screws are specifically designed to address these needs, slowly degrading as the natural tissue takes over, thereby eliminating the need for a second surgery to remove the implant.
Furthermore, the industrial trend in New Zealand is leaning toward sustainable and ethically sourced medical devices. Yuna Medical’s commitment to "Expanding with love, Striving for progress" aligns with the Kiwi ethos of care and quality. Our manufacturing processes utilize precision CNC machining and rigorous quality control to ensure that every screw—whether it is a SS304 Knurled Thumb Screw for equipment or a Bioabsorbable ACL screw—exceeds international medical standards.
For New Zealand distributors and hospital procurement officers, understanding the technical nuances of interference screws is critical. Our product range covers a variety of diameters and lengths to accommodate diverse patient anatomies. The Grab Slotted Headless design provides a low-profile finish that prevents soft tissue irritation—a common concern in thin-skinned areas of the knee and ankle.
In the industrial sphere, we also cater to specialized medical equipment requirements. Our Non-Magnetic 316L Stainless Steel Standoff Screws are essential for New Zealand’s growing network of MRI and diagnostic imaging centers. By eliminating electromagnetic interference, these components ensure that precision calibration and high-fidelity imaging are maintained, even in the most sensitive laboratory environments in Wellington and Dunedin.
Exporting to New Zealand requires a deep understanding of logistics and sterile packaging. Yuna Medical ensures that all orthopedic implants are double-sterilized and packaged in validated clean-room environments. We provide comprehensive documentation for Medsafe NZ registration, making the import process seamless for our Kiwi partners. Whether you are looking for high-quality wholesale ACI cannulated screws or bespoke orthopedic solutions, our 30 years of manufacturing expertise makes us the preferred choice in the South Pacific.
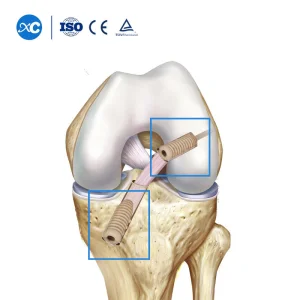 Non Absorbable Cannulated Peek Interference Arthroscopy Orthopedic Screws
Non Absorbable Cannulated Peek Interference Arthroscopy Orthopedic Screws
 High-Quality Wholesale Aci Cannulated Interference Screws
High-Quality Wholesale Aci Cannulated Interference Screws
 Biocomposite Surgical Acl and Pcl Reconstruction Non Absorbable Screws Peek Interference Bone Screw for Soft Tissue Fixation
Biocomposite Surgical Acl and Pcl Reconstruction Non Absorbable Screws Peek Interference Bone Screw for Soft Tissue Fixation
 Orthopedic Products Supplier Acl Cannulated Interference Screw
Orthopedic Products Supplier Acl Cannulated Interference Screw
 Sports Medicine Interference Screws for Acl Reconstruction Peek and Cannulated Screws
Sports Medicine Interference Screws for Acl Reconstruction Peek and Cannulated Screws
 Acl Interference Screw for Knee Arthroscopy Peek Screw Interference Screw Instrument Set
Acl Interference Screw for Knee Arthroscopy Peek Screw Interference Screw Instrument Set
 SS304 Carbon Steel Knurled Head Thumb Screw
SS304 Carbon Steel Knurled Head Thumb Screw
 M20X90 Nylon Hex Screw Peek Bolt Fastener Hex Bolts
M20X90 Nylon Hex Screw Peek Bolt Fastener Hex Bolts
Join the network of New Zealand surgeons and healthcare providers who trust Yuna Medical for their critical fixation needs. Our interference screws represent the pinnacle of medical engineering, designed to restore mobility and enhance life quality.
Send Inquiry Now