Chicago, a powerhouse of medical research and industrial manufacturing, has emerged as a critical epicenter for the Maxillofacial Implants industry. As a city home to prestigious institutions like Northwestern Memorial Hospital and the University of Chicago Medical Center, the demand for precision-engineered Craniomaxillofacial (CMF) solutions is at an all-time high. Our manufacturing facilities serve the growing needs of surgeons across the Illinois Medical District, providing advanced titanium and PEEK (Polyether ether ketone) implants that redefine reconstructive surgery.
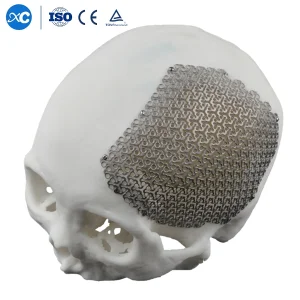
The Maxillofacial Implants Manufacturer & Factories in Chicago landscape is characterized by a unique blend of "Old School" precision machining and "New Era" additive manufacturing. Chicago’s industrial heritage provides the foundation for high-tolerance metalwork, while its burgeoning biotech sector drives the adoption of 3D-printed, patient-specific implants (PSIs). This synergy allows for the production of everything from standard micro-plates to complex orbital floor reconstructions that fit a patient's anatomy with sub-millimeter accuracy.
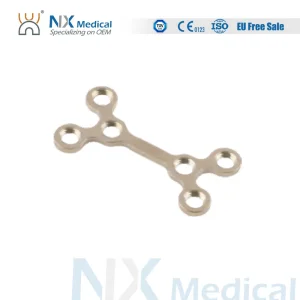
In the heart of the Midwest, surgeons are increasingly looking for Variable Angle Locking Compression Plates (VA-LCP). These systems allow for multidirectional screw insertion, which is crucial when dealing with complex fractures of the mandible or midface where bone quality may be compromised. Our Chicago-linked factories specialize in these high-tech systems, ensuring that local trauma centers have immediate access to the latest orthopedic technology.
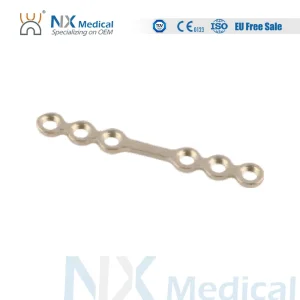
Furthermore, the shift toward minimally invasive surgery in Chicago’s outpatient clinics has spurred the development of micro-plating systems. These systems utilize ultra-low profile plates and screws that reduce palpable hardware and improve patient comfort post-surgery. Whether it is for elective plastic surgery or emergency trauma reconstruction, the CMF solutions originating from our specialized production lines meet the rigorous standards of the FDA and the high expectations of the American surgical community.
The move toward Patient-Specific Implants (PSI) is the most significant trend in Chicago. Using CT data, we manufacture titanium mesh and plates that perfectly match the patient's unique skull geometry, reducing operating time by up to 30%.
PEEK is becoming the material of choice for cranial repairs due to its radiolucency and bone-like elasticity. Our factories are at the forefront of machining medical-grade PEEK for Chicago’s leading neurosurgical departments.
Advanced surface treatments, such as anodic oxidation and hydroxyapatite coating, are being applied to titanium implants to enhance osseointegration and reduce the risk of implant rejection in complex reconstructions.
Changzhou Yuna Medical Apparatus And Instruments Co., Ltd. (hereinafter referred to as Yuna Medical) was established in 1992. As a high-tech enterprise integrating research, development, manufacturing, and sales, Yuna Medical has maintained steady growth for the past 30 years and has entered a phase of rapid expansion since 2017.
Guided by the spirit of “Expanding with love, Striving for progress,” Yuna Medical is committed to technological innovation and the development of advanced medical products that empower clinical professionals and benefit patients worldwide. Our main product lines include Orthopedic Power Tool Systems, Orthopedic External Fixation Systems, and specialized Maxillofacial Implants designed for international markets, including the demanding medical corridors of Chicago and the broader United States.

With three decades of mastery, Yuna Medical continuously advances in orthopedic technology and global market presence, ensuring Chicago hospitals receive tested solutions.

Over 20% of our workforce are R&D professionals driving innovation in orthopedic and rehabilitation solutions, focusing on the future of CMF surgery.

Certified by ISO, CE, FDA, and CCC, ensuring our Chicago clients receive top-tier product quality that meets all US healthcare regulations.

Collaborating with leading universities and surgeons in Illinois and beyond to enhance research, product development, and clinical effectiveness.



The manufacturing process of maxillofacial implants in the Chicago region involves sophisticated CNC machining and Vacuum Melting techniques. Titanium Grade 5 (Ti-6Al-4V ELI) is the primary material used due to its exceptional strength-to-weight ratio and bio-inertness. In our Chicago-affiliated factories, we employ multi-axis milling machines to create plates with thicknesses as thin as 0.6mm, catering to delicate pediatric craniofacial surgeries.
One of the localized application scenarios involves the high incidence of facial trauma in urban centers. Trauma surgeons at Chicago’s Level 1 trauma centers require Maxillofacial Locking Micro/Mini & Reconstruction Implants that can be contoured in the operating room without losing structural integrity. Our "Chicago Tough" series of plates are designed with a specific grain structure that allows for anatomical bending while preventing stress corrosion cracking.
As the Maxillofacial Implants Manufacturer & Factories in Chicago market evolves, we are investing heavily in "Smart Implants" that incorporate biosensors to monitor bone healing and detect early signs of infection. This AI-driven approach aligns with Chicago's position as a tech-forward city, bridging the gap between hardware and digital health.