
1 / 5










| Customization: | Available |
|---|---|
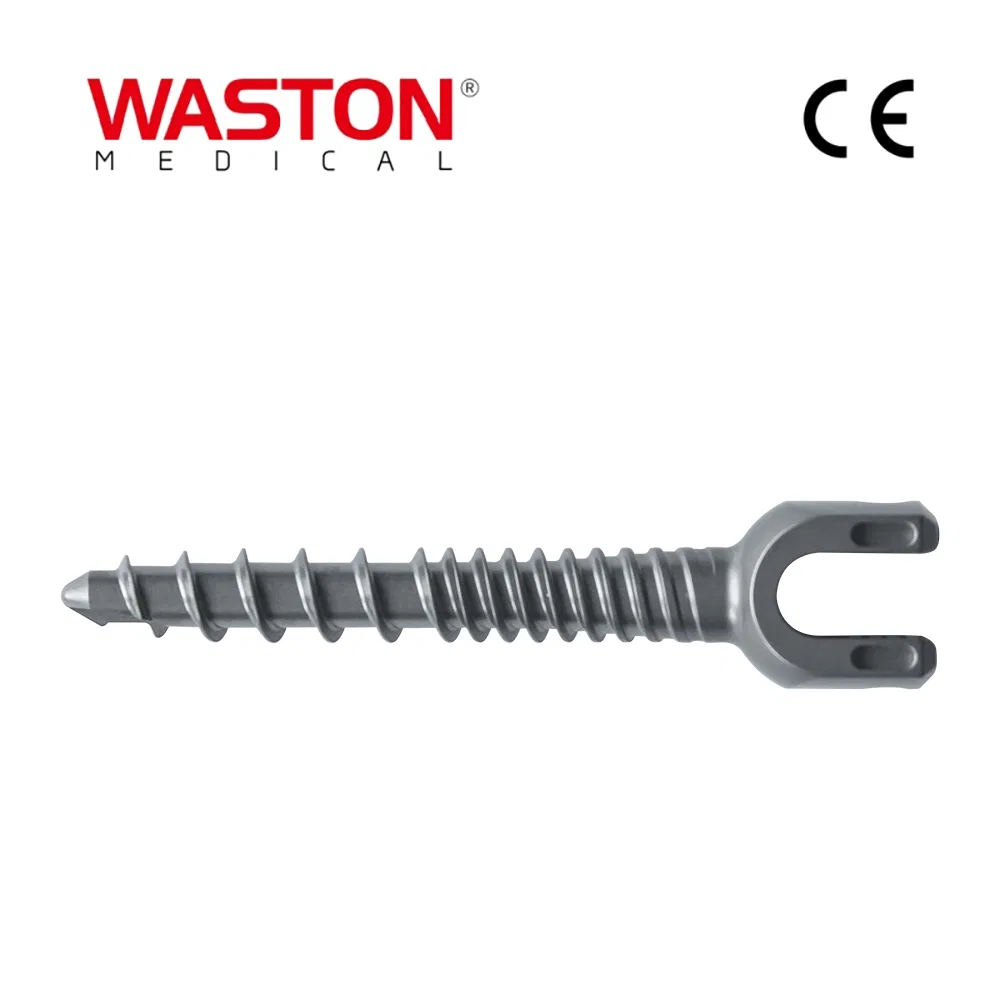
| Type: | Surgical Needle & Hook |
| Application: | Orthopedic |
| PRODUCT NAME | PRODUCT CODE | DIA | DATA |
|---|---|---|---|
| Laminoplasty Plate | 11101012 | / | H=12 |
| 11101014 | / | H=14 | |
| 11101016 | / | H=16 | |
| Screw (Cross self-tapping) | 10511306 | φ2.5 | L=6 |
| 10511308 | L=8 | ||
| 10511310 | L=10 |


As a national high-tech enterprise, the proportion of R&D investment to total sales has been over 8% for consecutive years. This facilitates the transformation of clinical experience and innovative ideas into achievements, launching several products with national invention patents.

The enterprise operates strictly according to the ISO13485 quality system, adhering to medical device manufacturing quality management specifications and CE MDD 93/42/EEC directives. High-end processing equipment from Germany, America, and Japan ensures first-class quality.
Products are exported to numerous countries including Columbia, France, Turkey, Indonesia, Mexico, Russia, and Thailand. Feedback confirms that the quality meets international standards.



 Wigivida Medical
Wigivida Medical