Basic Info.
Model NO.
External Fixation
Certification
CE, ISO13485
Material Type
Titanium Alloy & Stainless Steel
Capacity
500,000 Pieces/Year
Product Description
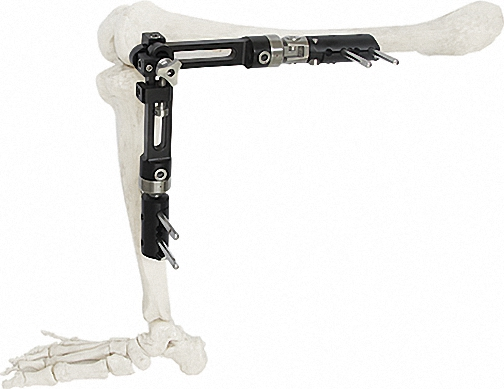
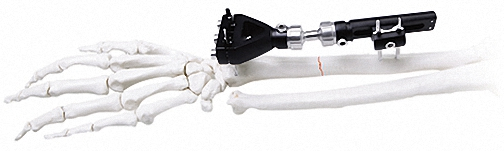
External Fixator -- The Pinnacle of Modular Fixation Systems
Product Features
- Groundbreaking technology and intuitive, user-friendly operation.
- Facilitates independent arrangement of bone screws for proximal and distal fractures.
- Innovative snap-fit design for streamlined and time-efficient surgical procedures.
- Premium Medical aluminum and carbon fiber materials for superior fracture visualization.
- Engineered for unmatched stability and flexibility across diverse medical needs.
- Compact, space-saving frame for ease of use and transport.
- Designed to manage complex fractures and soft tissue loss.
| Component Name |
Specification |
Quantity |
| Independent Coupling | φ6*φ14(1.5-2.0) / (1.5) | 10 / 10 |
| Needle Clip | φ4*φ9 (φ1.0) / (φ1.5) | 5 / 5 |
| Cone Washer | φ4*φ9 | 10 |
| Double Groove Needle Clamp | φ4*φ14(2.0-2.5) | 5 |
| Connection Plate | φ4 (2-5 Holes) | 4 each |
| Post With Threaded Post | Various (φ3-φ4) | 8 each |
| Hex Nut | M3 / M4 | 90 / 90 |
| Bone Screws | 1.0 / 1.2 / 1.5 * 150 | 20 / 20 / 8 |
Specialized Fixator Models
Lower Limbs Lengthening External Fixator
Used for Tibia and Femur procedures.
Orthopedic External Fixator
Used for Humeral & Tibial Lateral Epicondyle.

Ring Fixator (Tibia/Femur)

2/3 Ring Fixator

Ankle Joint Fixator

Knee Joint Fixator
Frequently Asked Questions
What is the primary material used in these external fixators?
The fixators are primarily constructed from high-grade Titanium Alloy and Stainless Steel. Some models also utilize Medical Aluminum and Carbon Fiber to improve X-ray visualization.
Are these devices reusable?
Yes, the core External Fixation system components are designed to be reusable after following proper sterilization and maintenance protocols.
Which types of fractures can be managed with this system?
This modular system is versatile enough to handle proximal and distal fractures, complex multi-fragment fractures, and cases involving significant soft tissue loss.
What certifications do these medical devices hold?
Our external fixator products are fully certified with CE and ISO13485 standards, ensuring compliance with international medical quality requirements.
How does the snap-fit design benefit the surgical process?
The innovative snap-fit design allows for quicker assembly and adjustments during surgery, significantly reducing the overall operation time and improving surgeon efficiency.
What is the typical production capacity for these units?
The production facility has a high-volume capacity of approximately 500,000 pieces per year to meet global orthopedic demands.
























 Wigivida Medical
Wigivida Medical