











| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |


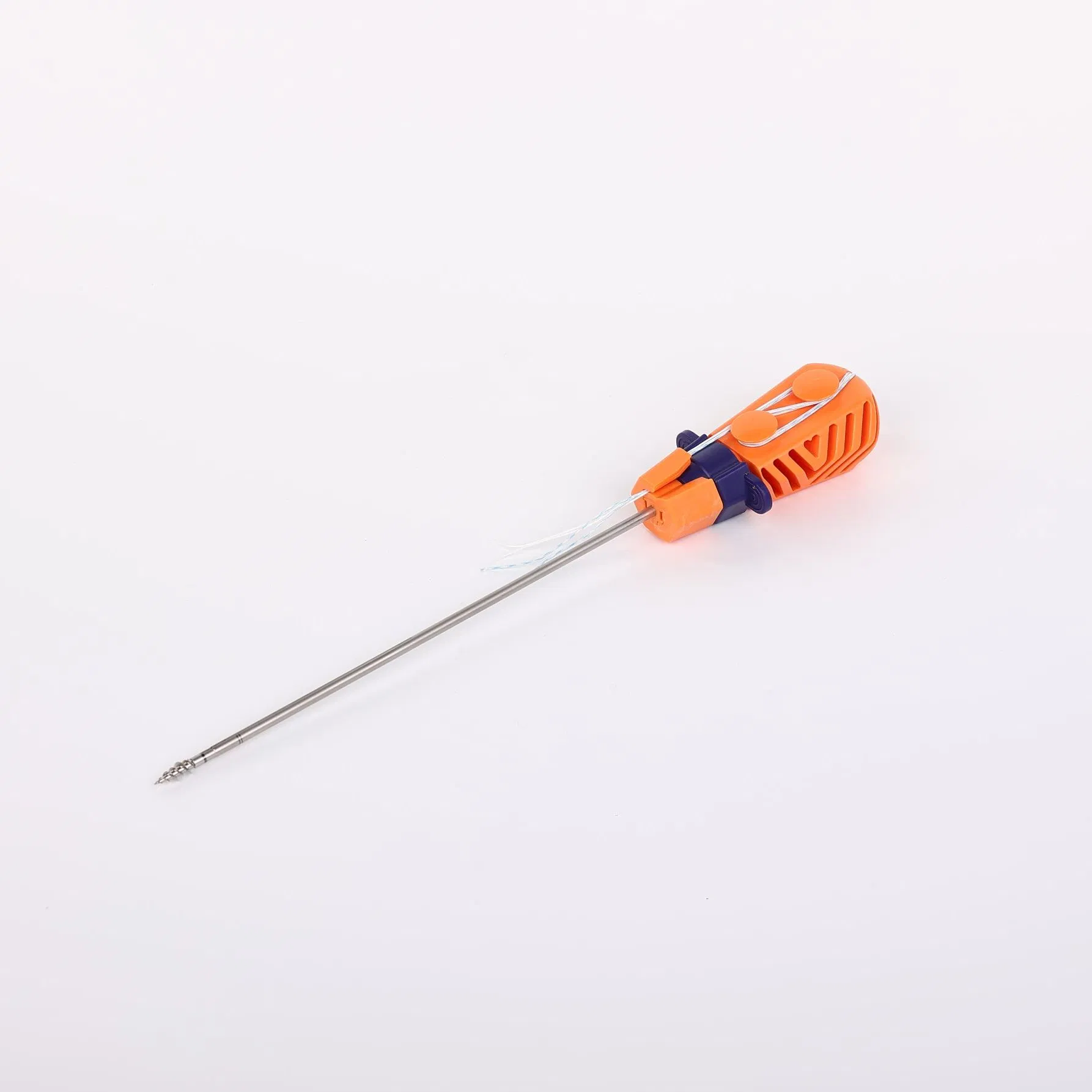
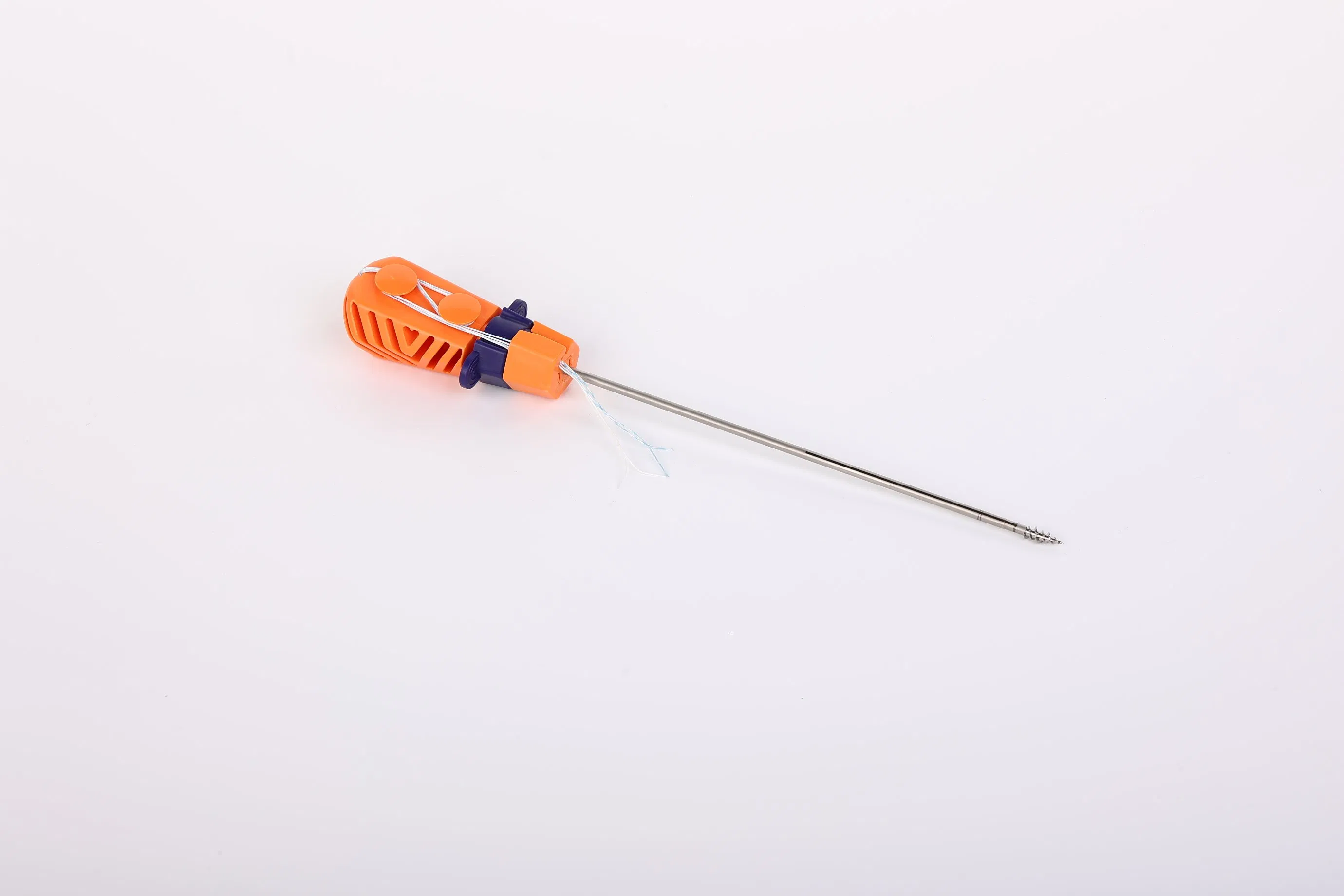
The Suture Anchor System is a precision-engineered orthopedic solution consisting of an anchor, non-absorbable suture, and an inserter. The anchor is fabricated from high-grade Ti6Al4V alloy, strictly adhering to ISO 5832-3:2016 standards for biocompatibility and strength.
The inserter features a stainless steel tip (ASTM F899-12b) for surgical precision, coupled with an ergonomic handle made of durable polycarbonate and ABS material, designed for optimal control and ease of use.

Our manufacturing process utilizes advanced Class 100,000 clean workshops and Class 10,000 purification laboratories. This ensures every orthopedic consumable, from meniscal repair systems to suture anchors, meets international safety benchmarks including CE and ISO 13485 certifications.




 Wigivida Medical
Wigivida Medical