📋 Basic Information
Certification
CE, ISO13485
Usage
Bone Fracture Fixation
Production Capacity
1000PCS/Month
🌟 Our Advantages
✓
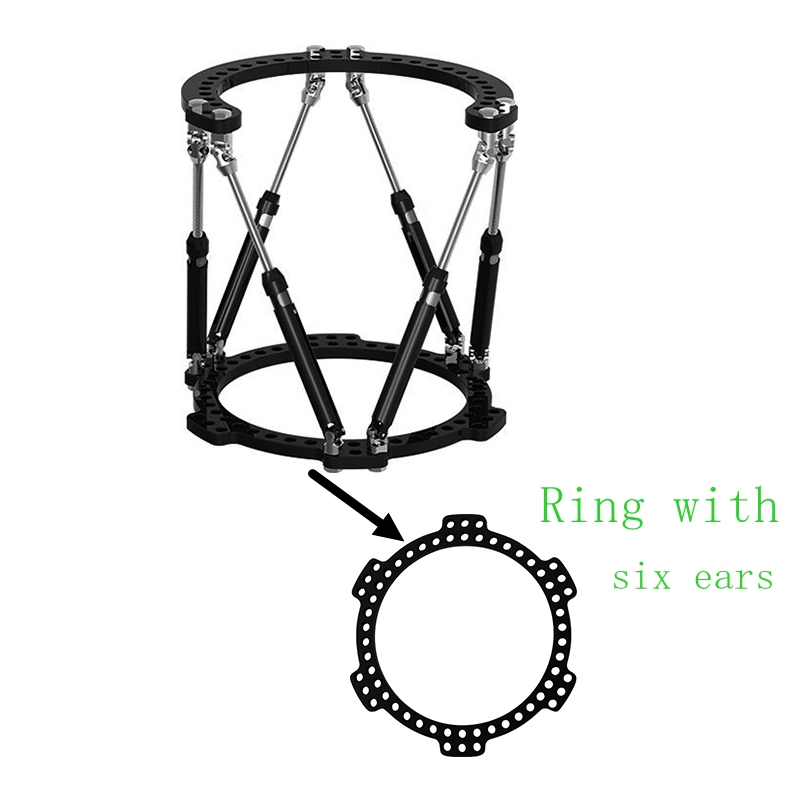
Annular design, firm and reliable
✓
Easier operation & shorter surgical time
✓
Minimally invasive Surgery, no effect to blood supply of bone
✓
Second surgery unnecessary, remove in clinic
✓
Dynamic design better for bone healing
✓
Taper bone screws, taut and firm after insertion
✓
Suitable for all kinds of deformity correction
📊 Product Specifications
| Name |
Number |
Size |
Material |
| Rings with 6 ears |
HTLL0101 |
L |
Aluminum |
| HTLL0102 |
M |
| HTLL0103 |
S |
📷 Detailed Photos
📜 Certifications
🏛 Exhibition
🏭 Profile Overview
Established in 1958, the company is engaged in the development, manufacture and sales of orthopedic implants and instruments. As a key enterprise in the medical apparatus industry, the product line covers Spine pedical screws, Interlocking nails, Locking plates/screws, trauma plates/screws, cannulated screws, external fixators, hip prosthesis and surgical instruments.
The products are characterized by multi-varieties and complete specifications. All medical apparatus have passed the approval and authentication of national and provincial superintendent offices. Advanced equipment including imported CNC, high precision digital control machines, and 100,000 Grade axenic purification chambers contribute to the high quality of the products.
The company was among the first to pass ISO 9001 quality system certification. The products have won various national invention prizes and science and technology awards, establishing a reputable presence in the medical field.
❓ Frequently Asked Questions
What are the main applications for this external fixator?
It is primarily used for bone fracture fixation and various types of orthopedic deformity corrections in adult patients.
How do you ensure the quality of your orthopedic implants?
Products are manufactured according to strict EU CE standards. We conduct pre-production sample testing and a final inspection before every shipment.
What materials are used for the rings?
The rings are available in high-quality Aluminum or Stainless Steel (Ss) to ensure both strength and biocompatibility.
Is the fixator removal process complicated?
No, the design allows for removal in a clinical setting, making a second major surgery unnecessary for the patient.
What is the production capacity and lead time?
Our current production capacity is approximately 1,000 pieces per month. Lead times depend on order size and shipping requirements.
Which shipping methods do you support?
We offer shipping via major international carriers including DHL, UPS, FedEx, EMS, and TNT Air Cargo.











 Wigivida Medical
Wigivida Medical