











| Still deciding? Get samples of $ ! US$ 15/Piece Request Sample |
| Products Name | NO. | Specification |
|---|---|---|
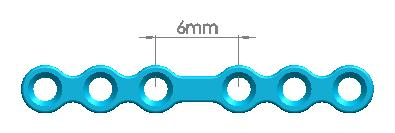
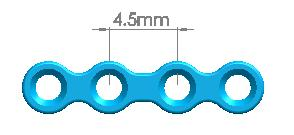
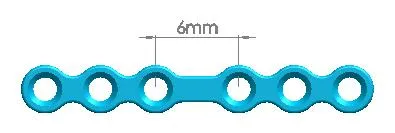
| Straight Maxillofacial Plate, Type-IV | 1500-0115 | 6 holes (Interspace: 6.0mm) |
| 1500-0116 | 6 holes (Interspace: 7.5mm) | |
| 1500-0117 | 6 holes (Interspace: 9.0mm) | |
| 1500-0118 | 6 holes (Interspace: 12mm) | |
| Use: 1.5mm Self-tapping Screw / 1.5mm Self-drilling Screw | ||

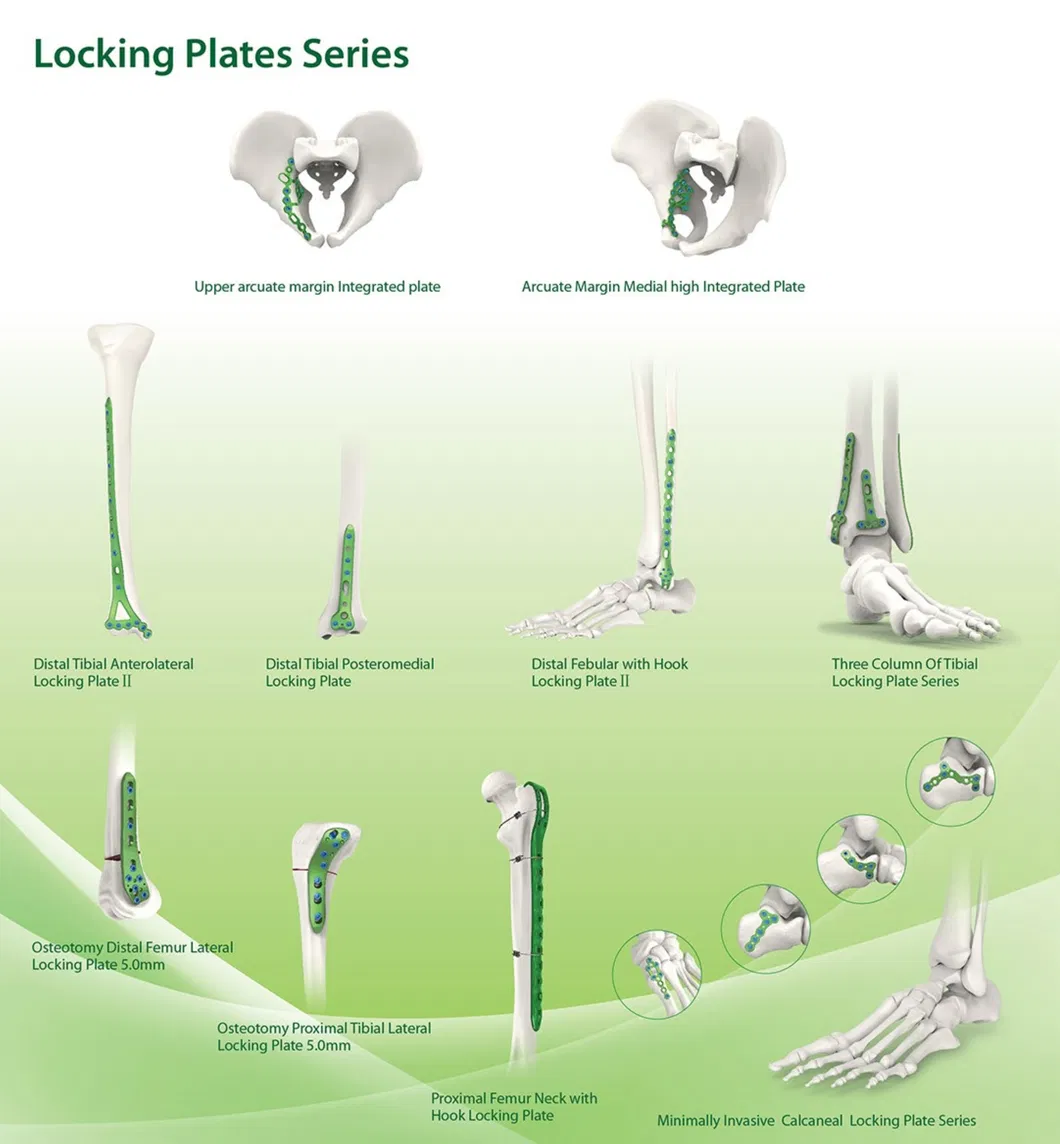






Established in 1958, the company is engaged in the development, manufacture, and sales of orthopedic implants and instruments. As a director company of the Medical Apparatus Industry Association, the product line covers spine pedicle screws, interlocking nails, locking plates/screws, trauma plates, cannulated screws, external fixators, hip prosthesis, and surgical instruments.
The products are characterized by multi-varieties and complete specifications. All medical apparatus have passed approval and authentication of national and provincial superintendent offices. Advanced equipment, including imported CNC, high-precision digital control machines, and milling machines, as well as a 100,000 Grade axenic purification chamber, contribute to high product quality.

 Wigivida Medical
Wigivida Medical