



1 / 5










| Still deciding? Get samples of $ ! US$ 20.65/Piece Request Sample |



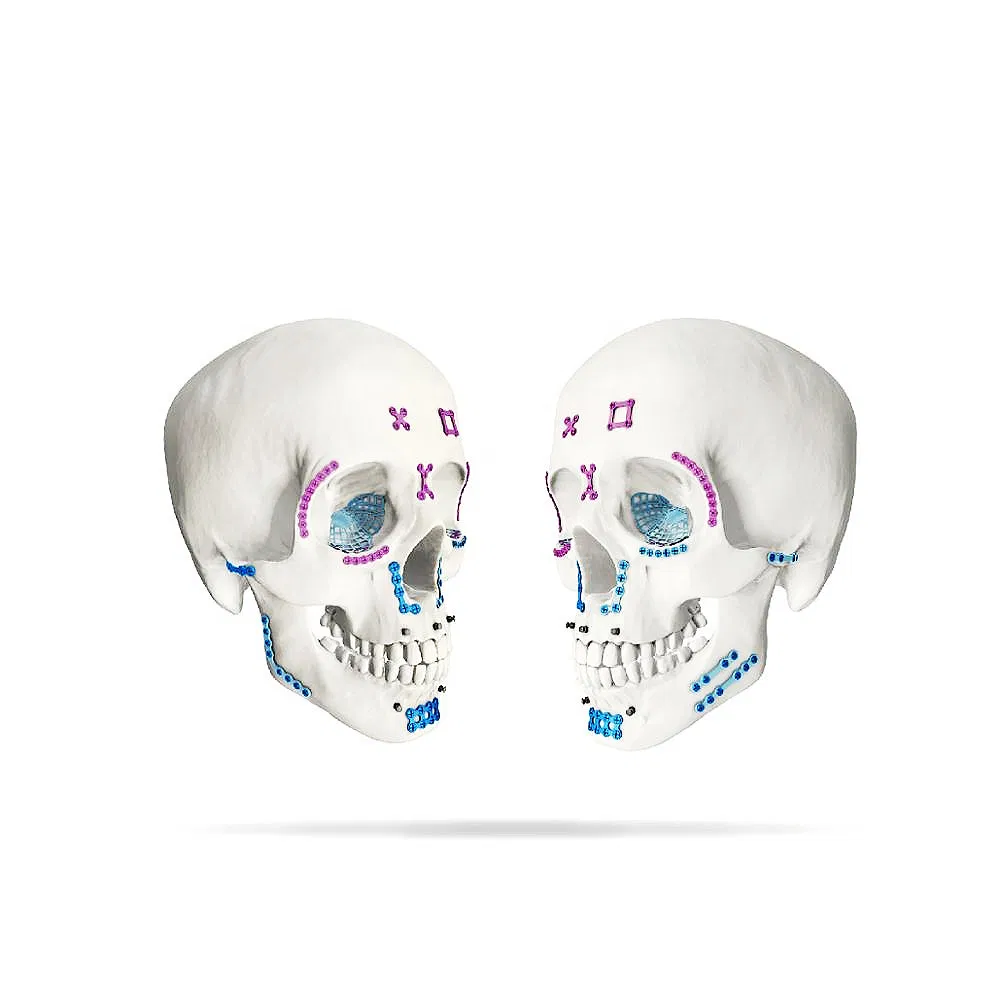
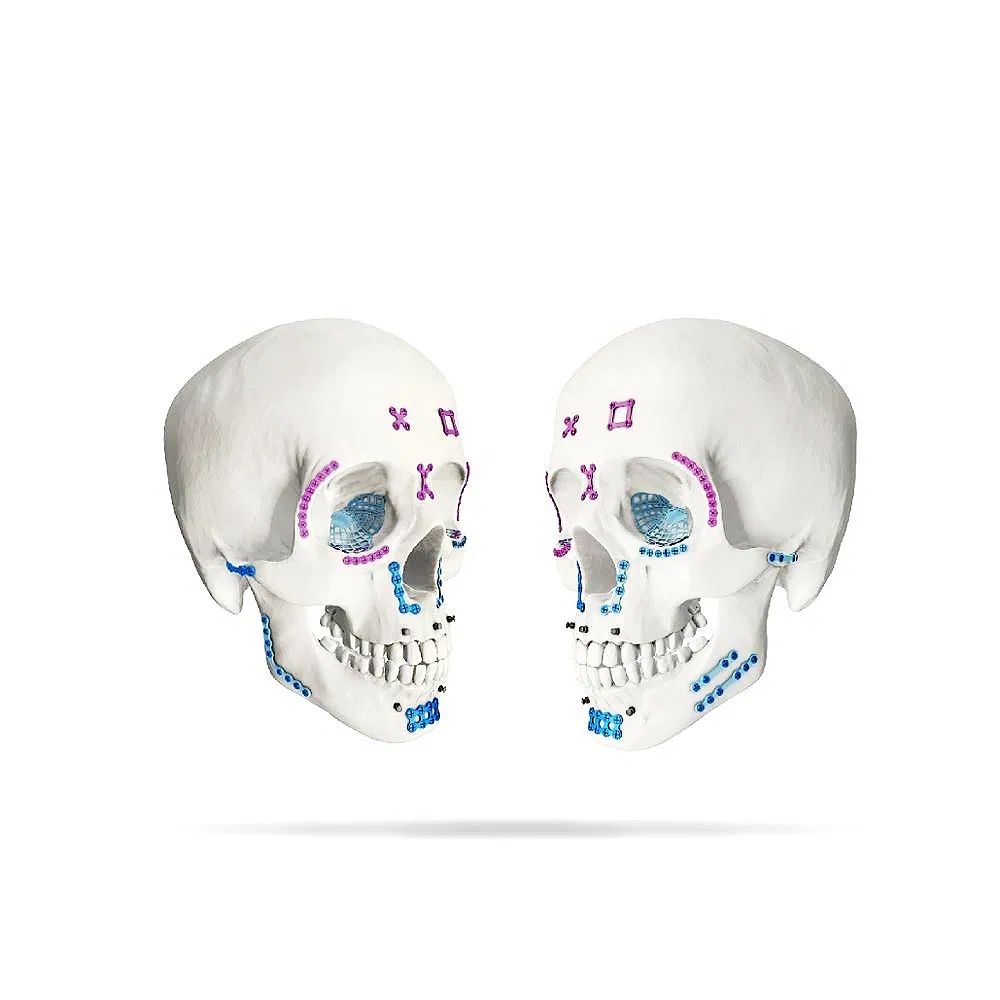
| Product Name | Orthopedic Medical Implant Maxillofacial Micro Titanium Straight Plate |
| Material | Pure medical titanium |
| Color | Purple / Customized |
| Specification | 4 / 6 / 8 / 10 / 12 / 14 Holes |
| Length | 16 / 24 / 32 / 40 / 48 / 56mm |
| Thickness | 0.6mm |
| Certification | CE / ISO9001 / ISO13485 |
| Indication | Front part, nasal part, pars orbitalis, pars zygomatica, maxilla region, pediatric craniofacial bone |







 Wigivida Medical
Wigivida Medical