











| Still deciding? Get samples of $ ! US$ 50/Piece Request Sample |

Related Products Showcase
Quality Certifications



Global Exhibition



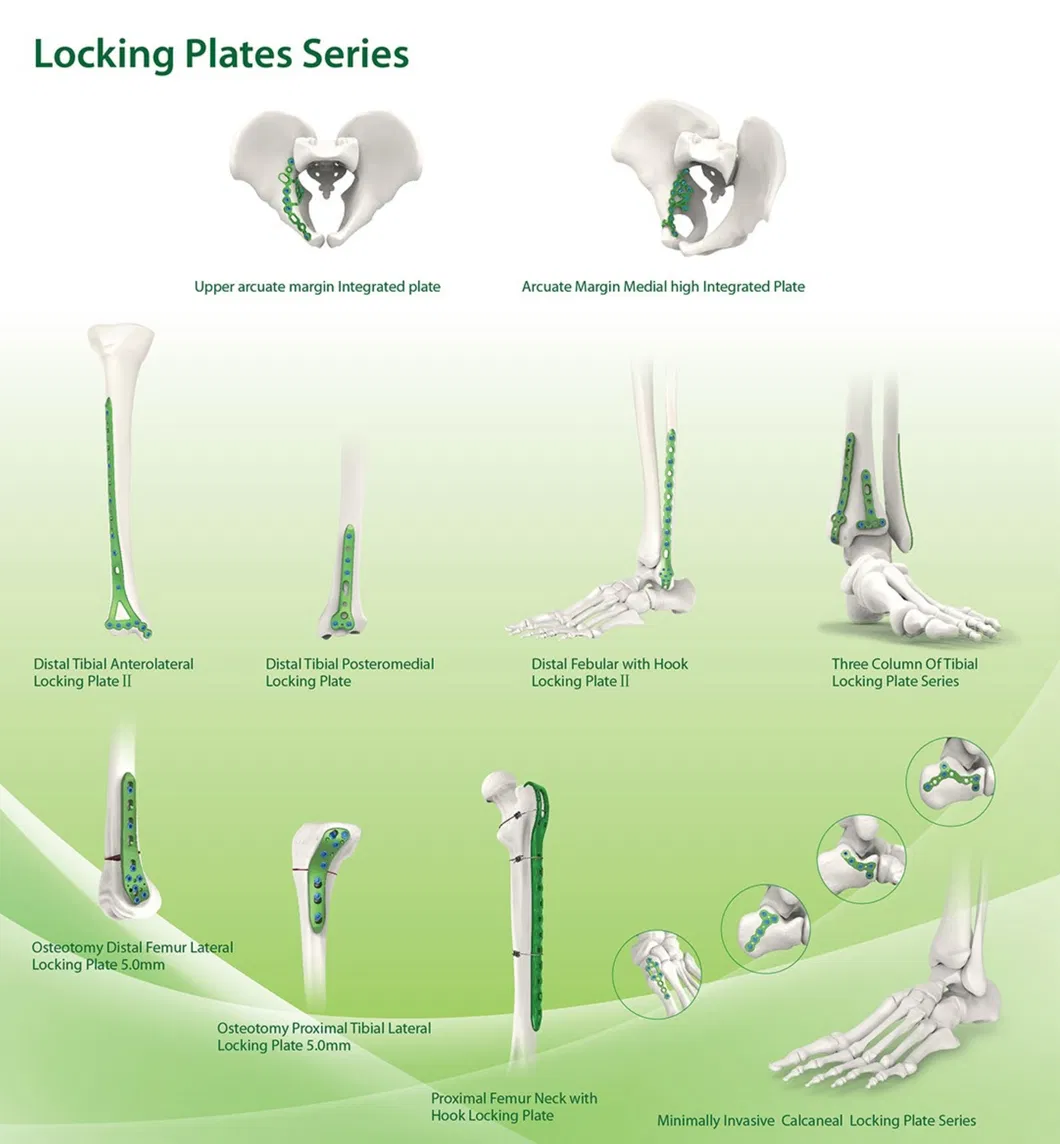
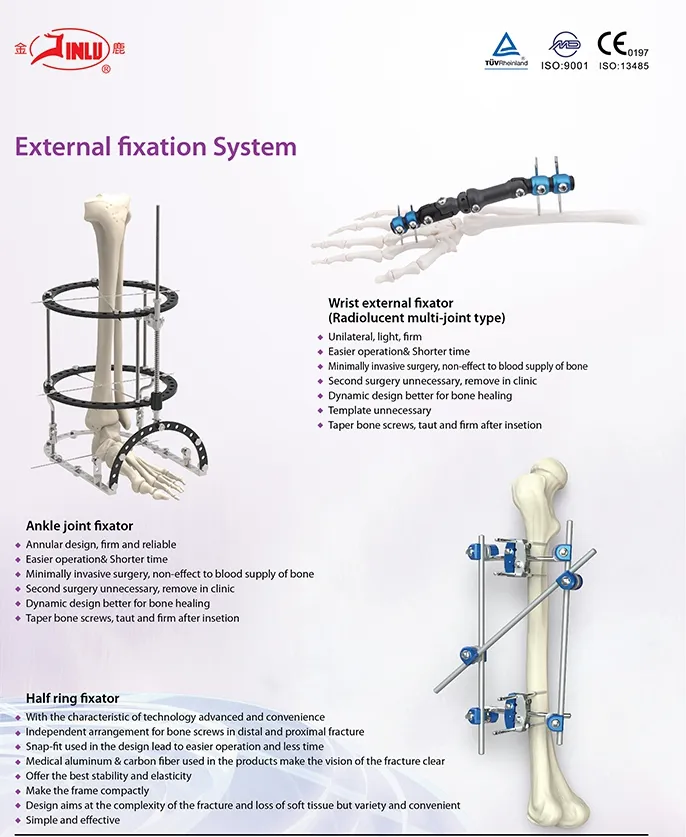
Established in 1958, we are dedicated to the development, manufacture, and sales of orthopedic implants and instruments. As a key enterprise in the medical apparatus industry, our extensive product line covers spine pedical screws, interlocking nails, locking plates/screws, trauma plates, cannulated screws, external fixators, hip prosthesis, and surgical instruments.
Our products are characterized by multi-varieties and complete specifications. All medical apparatus have passed the rigorous approval and authentication of national and provincial superintendent offices. Each product is marked with material codes and laser anti-fake marks for quality traceability.
Equipped with advanced imported CNC machines, high-precision digital control systems, and a 100,000 Grade axenic purification chamber, we ensure top-tier product quality. We were among the first to pass ISO 9001 quality system certification. Our commitment is to provide perfect designs, innovative technology, and comprehensive service to the global medical community.

 Wigivida Medical
Wigivida Medical