




1 / 5










| Still deciding? Get samples of $ ! US$ 32/Piece Request Sample |

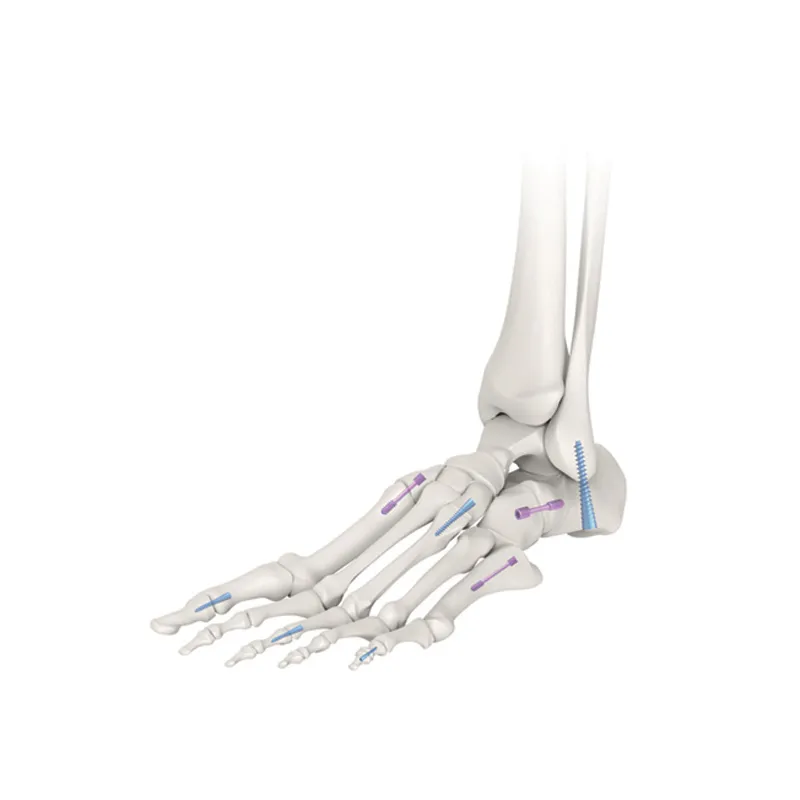
| P/N | Product Name | Material | Model/Specifications |
|---|---|---|---|
| 47235 | Cannulated Headless Compression Screw | Titanium Alloy Stainless Steel | 3.5*16/18~30mm |
| 47240 | 4.0*16/18~30mm | ||
| 47245 | 4.5*16/18~34mm | ||
| 47250 | 5.0*30/35~60mm |





 Wigivida Medical
Wigivida Medical