


1 / 5










| Still deciding? Get samples of $ ! US$ 1600/Set Request Sample |
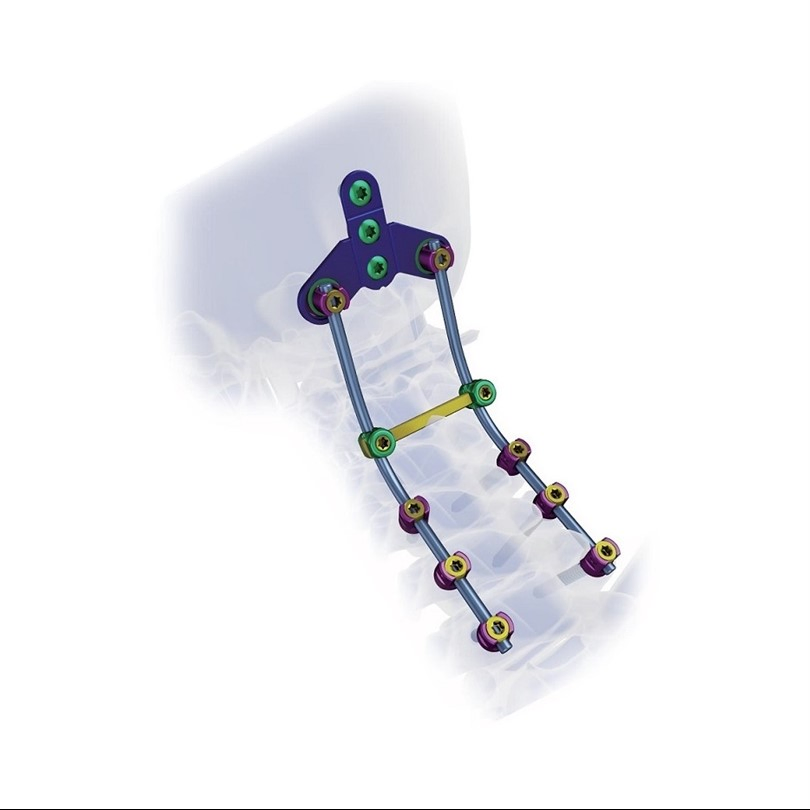
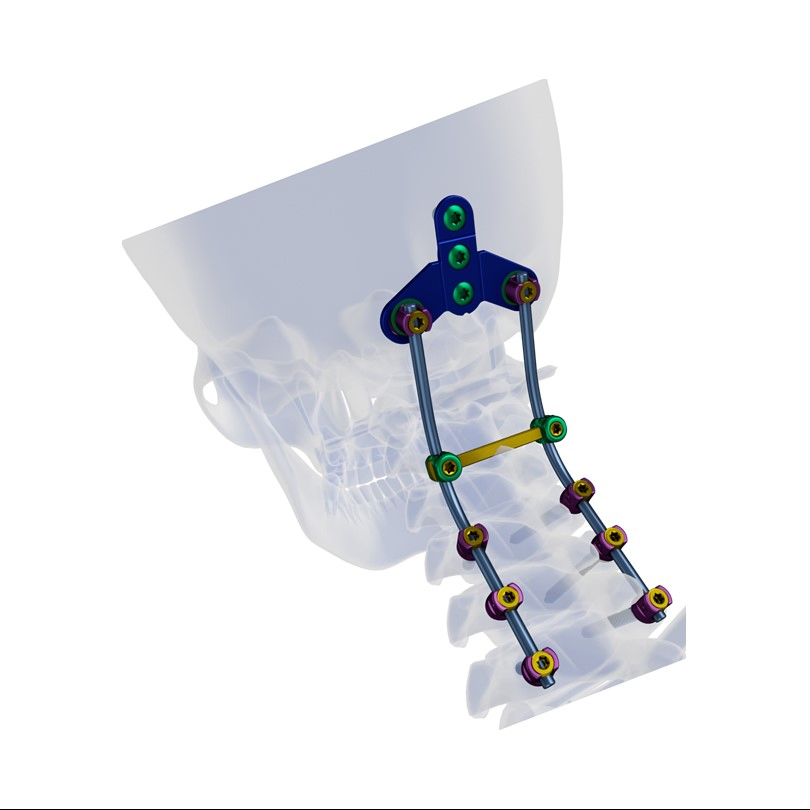
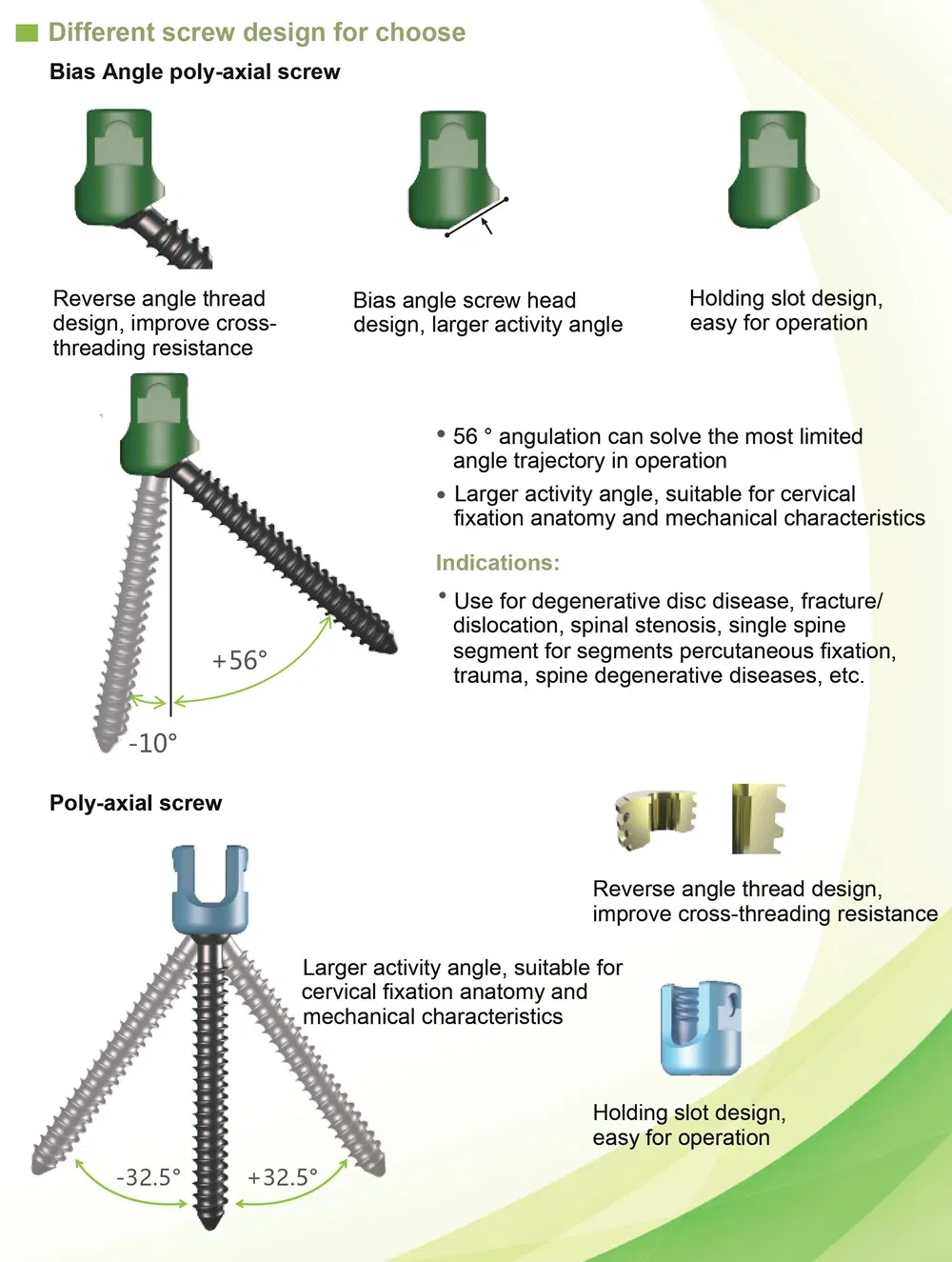
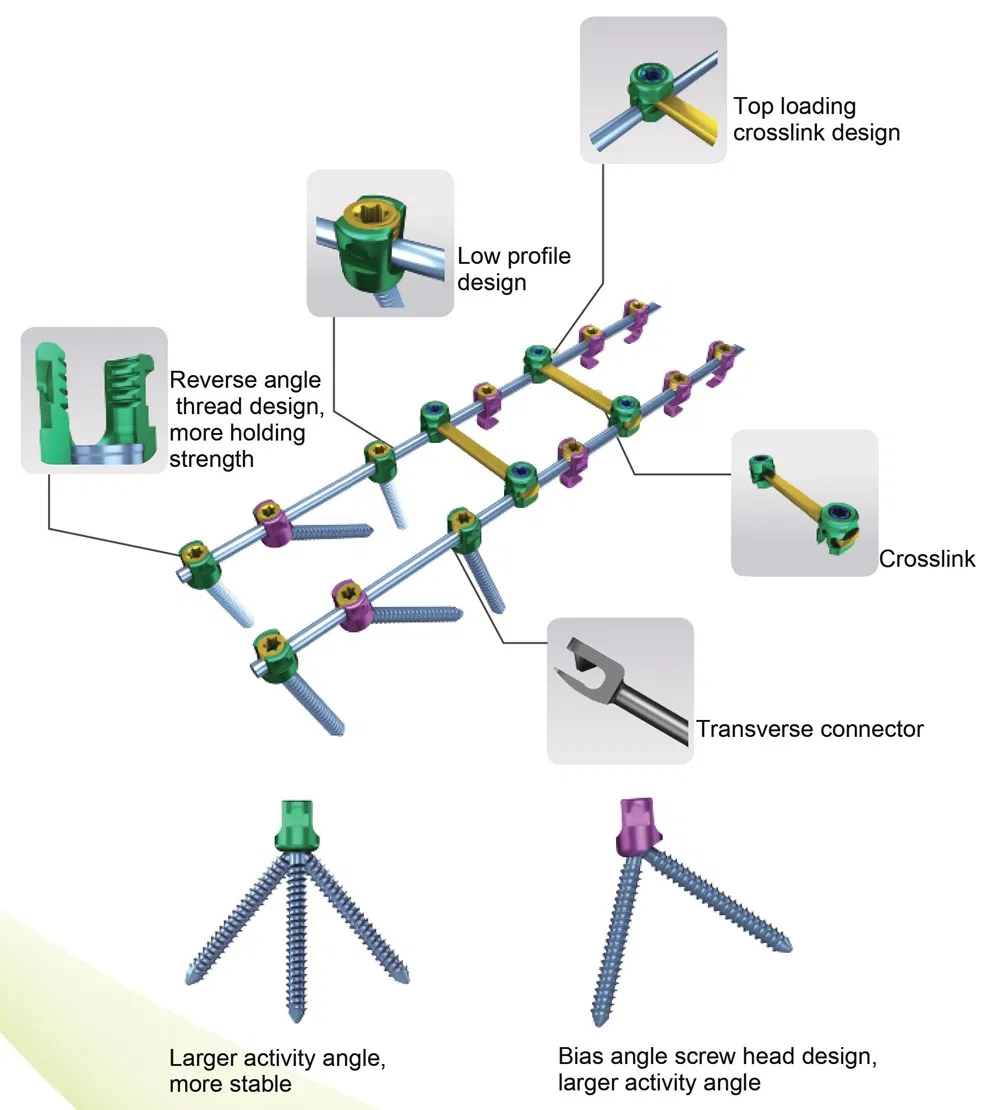
Function Description: Better biocompatibility, suitable for degenerative disc diseases, fracture / dislocation, spinal stenosis, occipital-cervical disassociation etc.
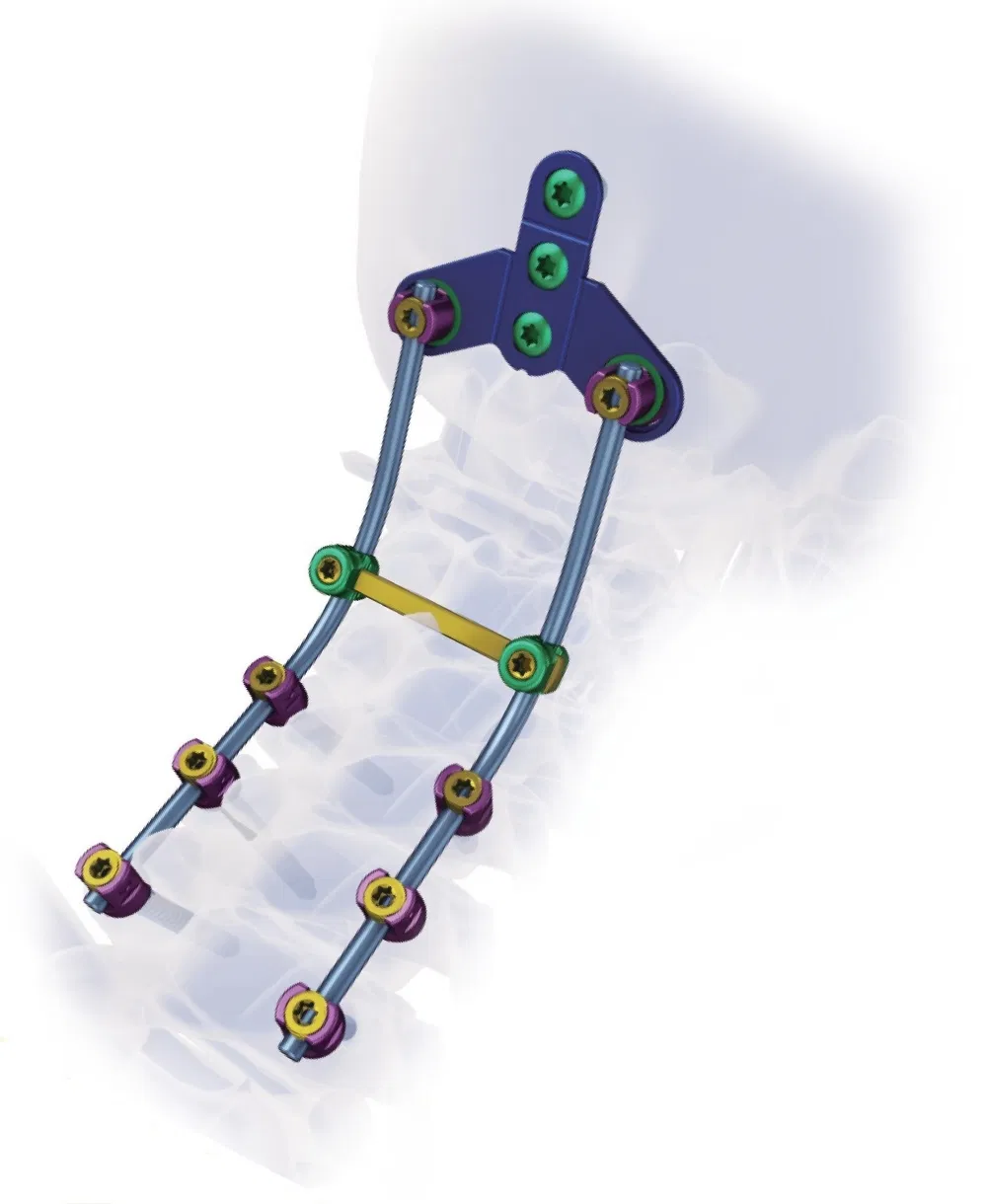


Packing Details:
Delivery Way: Reliable shipping via Air (DHL, UPS, FedEx, etc.).

Focus: One-stop orthopedic product solution provider since 2016.
Capacity: 10,000 units per month.
Global Reach: Products are exported to Southeast Asia, Middle East, Latin America, Africa, and Europe.

Factory Advantages:


 Wigivida Medical
Wigivida Medical