




1 / 5










| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |
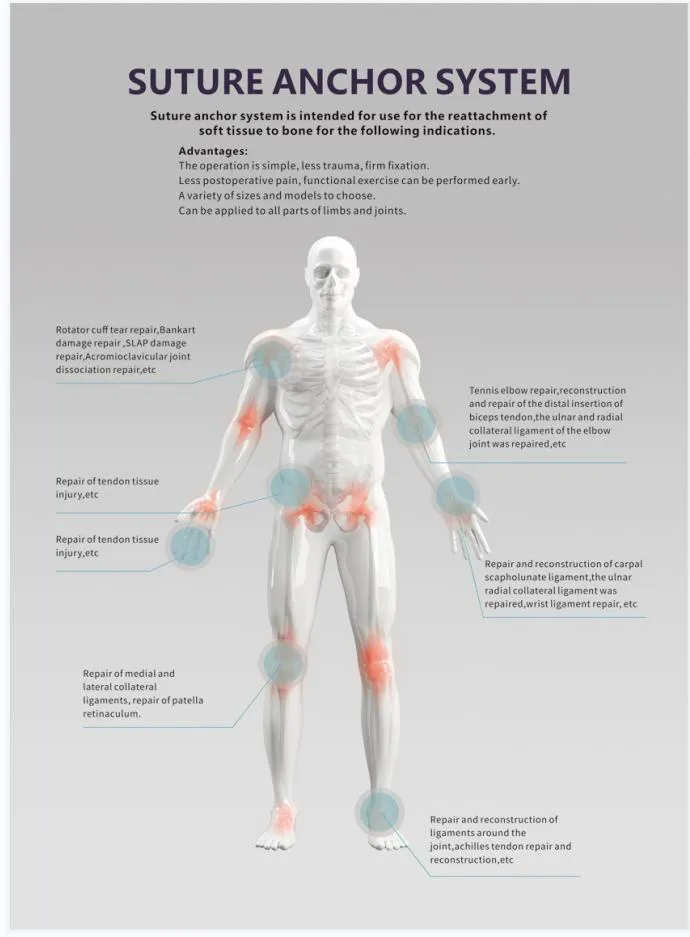
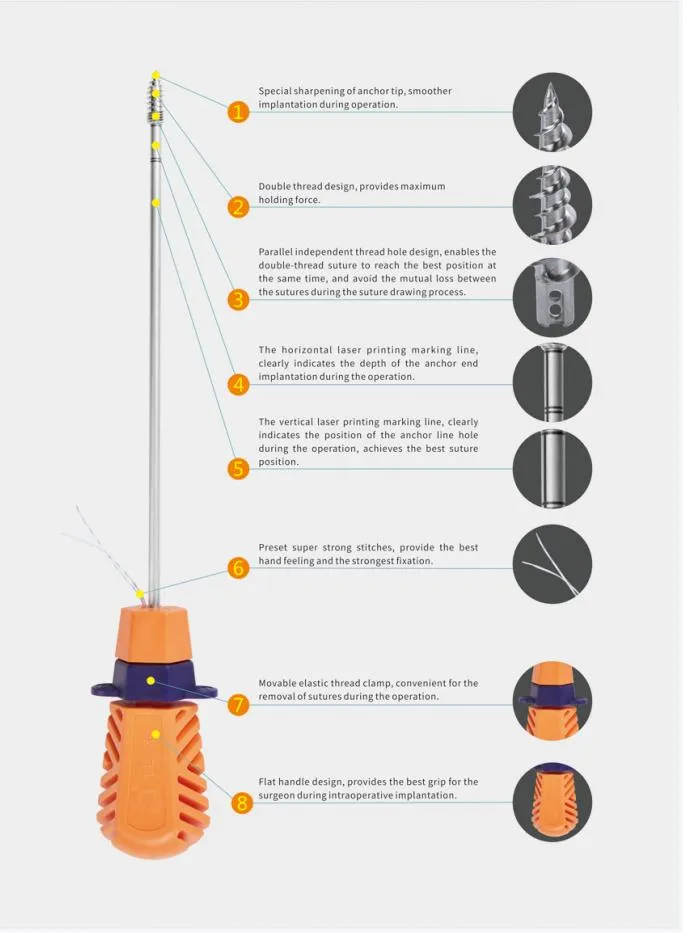
The Suture Anchor System for Sports Medicine Surgery is specifically designed for the repair and reconstruction of Rotator Cuff Tendons. This comprehensive system consists of a high-quality anchor, nonabsorbable suture, and an ergonomic inserter.







| Item Property | Detailed Specification |
|---|---|
| Product Name | Suture Anchor System |
| Material | Titanium Alloy / UHMWPE Suture |
| Classification | Class II |
| Dimensions | Diameter 1.8 x 6mm |
| Sterilization | EO Sterile Packaging |
| Application | Orthopedic Surgery / Implantology |
| Warranty | 1 Year (Parts) / 3 Years (Overall) |
 Wigivida Medical
Wigivida Medical