




1 / 5










| Still deciding? Get samples of $ ! US$ 100/Set Request Sample |








| Item | Value |
|---|---|
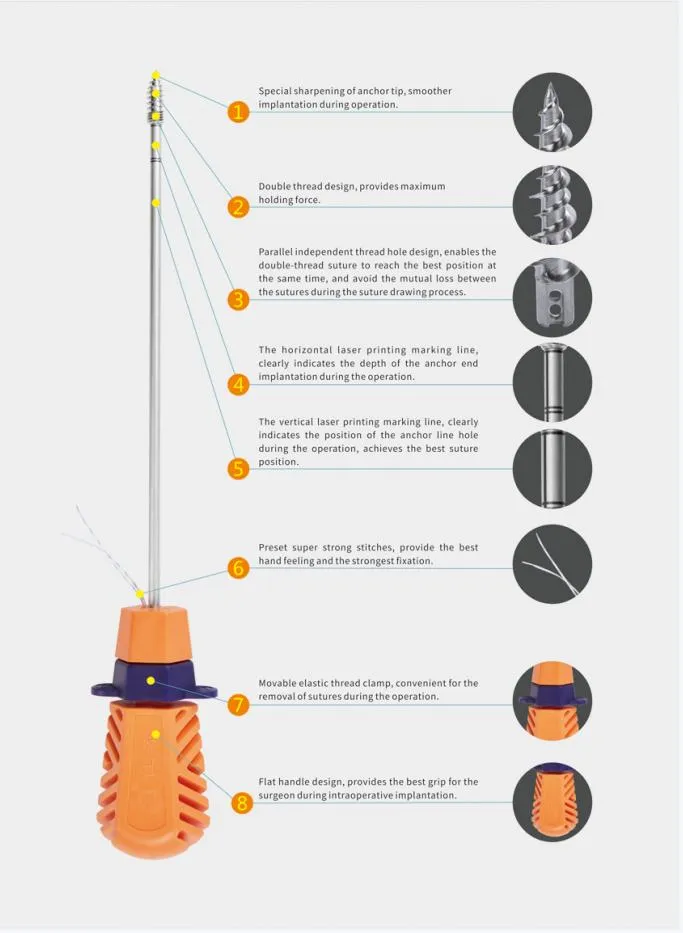
| Properties | Implant Materials & Artificial Organs |
| Model Number | WZ-MDT-S-01 |
| Instrument Classification | Class II |
| Warranty | 1 Year (After-sale) / 3 Years (Basic) |
| Material | Titanium Alloy |
| Color | Silver+Orange |
| Diameter Specification | 1.8x6mm |
| Package | Sterile Packaging 1pc/package |
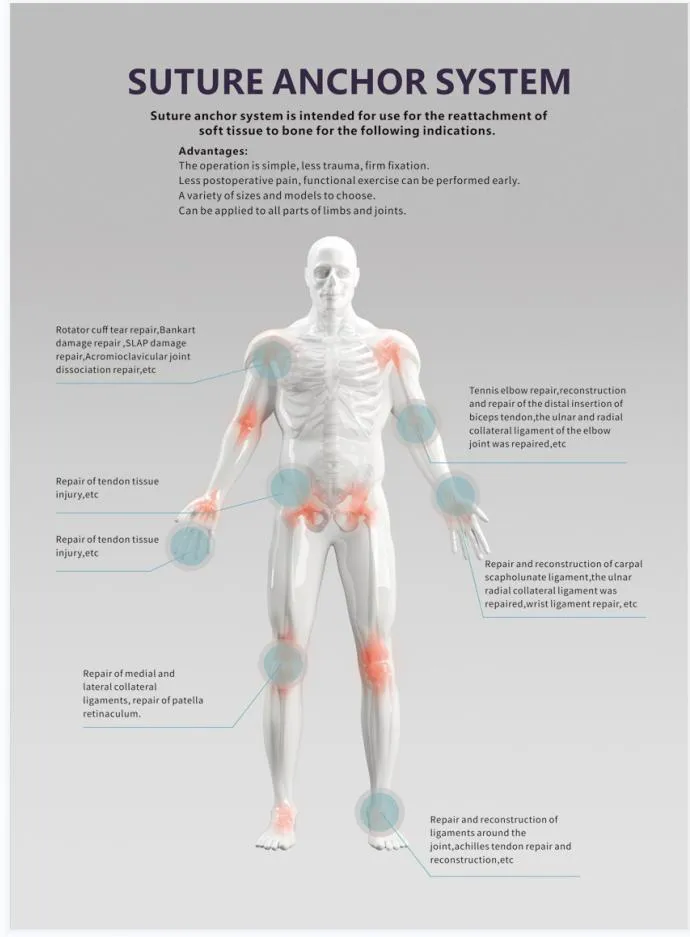
| Usage | Orthopedic Surgery |
| Application | Implantology |

 Wigivida Medical
Wigivida Medical