Using Site
Posterior Spinal Internal Fixation
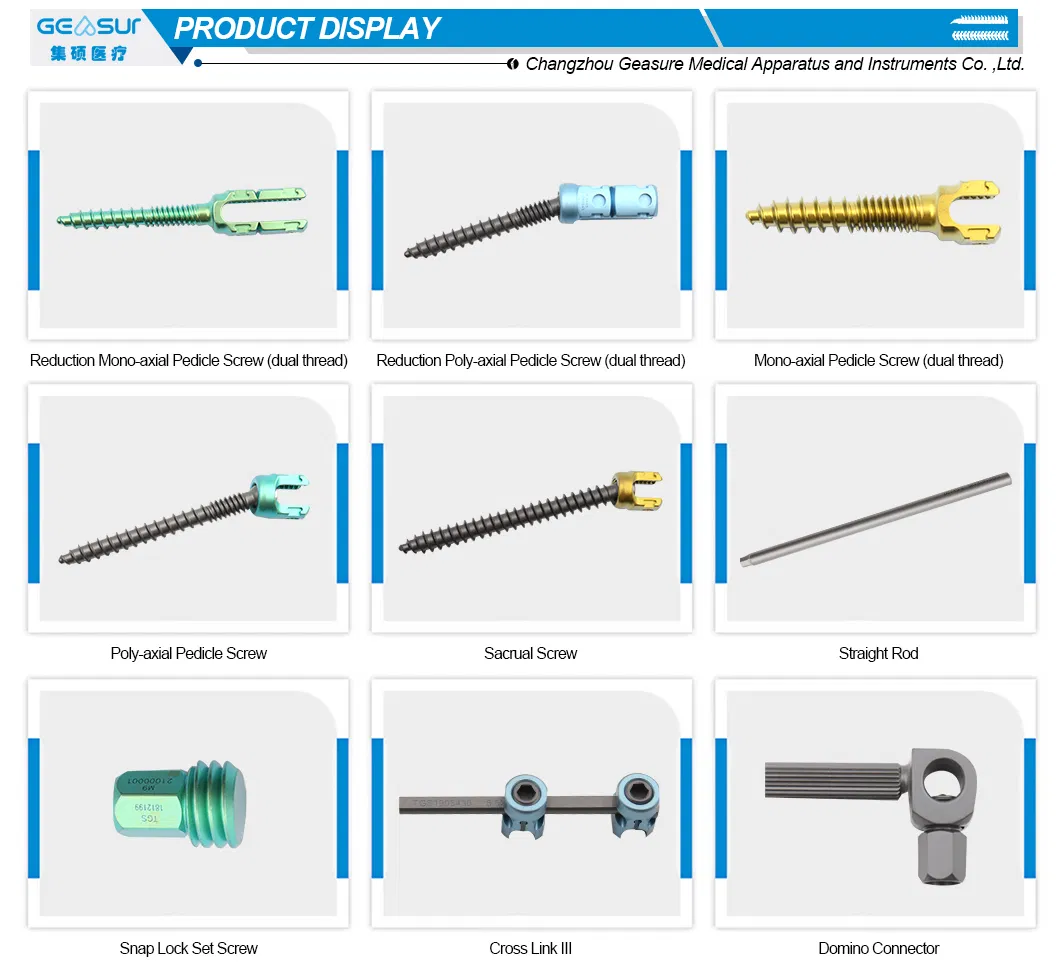
Product Name
Domino Connector
Instrument Class
Class III
Properties
Implant Materials & Artificial Organs
After-Sale
Return and Replacement
Transport
Standard Export Packaging
| Posterior Spinal Internal Fixation 5.5 System |
| Domino Connector (Material: Titanium) |
| Code (Non-Sterile) |
Code (Sterile) |
Diameter (mm) |
Length (mm) |
| 21005510 | 21085610 | 5.5 | 10 |
| 21005520 | 21085620 | 5.5 | 20 |
| 21005530 | 21085630 | 5.5 | 30 |
We are a company specializing in the research and production of orthopedic implants and instruments, including trauma, spine, joint, and sports medicine. The sales market covers the entire domestic region and many countries all over the world.
We have first-class production equipment, professional R&D, technology, and manufacturing teams. We have also established an orthopedic laboratory collaborated with many university professors and hospital surgeons to provide operational demonstrations.
We have obtained ISO13485 system certification and CE certification. Some products have been registered with the FDA 510k. Our efficient marketing and sales service strategies have been recognized by our partners. Our goal is "Quality is the foundation of life & Innovation is the road of development".
What are your shipping modes and typical charges?
Normally we send through couriers like DHL, FEDEX, UPS, TNT, or postal services upon specific request from clients. Shipping charges depend on volume and weight.
Do you hold international quality certifications?
Yes, we have obtained ISO13485 and CE approvals. Several of our products are also registered with the FDA 510k, ensuring high standards for medical usage.
What is the minimum order quantity (MOQ)?
We generally expect minimum orders to be at least US$500. However, we support smaller sample orders for initial market testing. Free samples can be provided, though freight costs are covered by the client.
Can you provide products not listed in the current catalogue?
Yes, if you provide pictures or physical samples of similar orthopedic products, we can accommodate your needs, though custom production may require additional lead time.
What are your payment terms and stock availability?
We maintain monthly stock for most listed products. For payments under $800, 100% advance is required. Above $800, we require 30% T/T in advance and the balance before shipment. We accept L/C, T/T, Western Union, and PayPal.
Do you provide after-sales support for your medical instruments?
We offer return and replacement services for our orthopedic implants. Our products come with a lifetime warranty to ensure long-term reliability for medical facilities.
























 Wigivida Medical
Wigivida Medical